When a heart attack strikes, heart muscle cells die and scar tissue forms, paving the way for heart failure.
Cardiovascular diseases are a major cause of death worldwide, in part because the cells in our most vital organ do not get renewed. As opposed to blood, hair, or skin cells that can renew themselves throughout life, our heart cells cease to divide shortly after birth, and there is very little renewal in adulthood. New research at the Weizmann Institute of Science provides insight into the question of why the mammalian heart fails to regenerate, on one hand, and demonstrates, in adult mice, the possibility of turning back this fate.
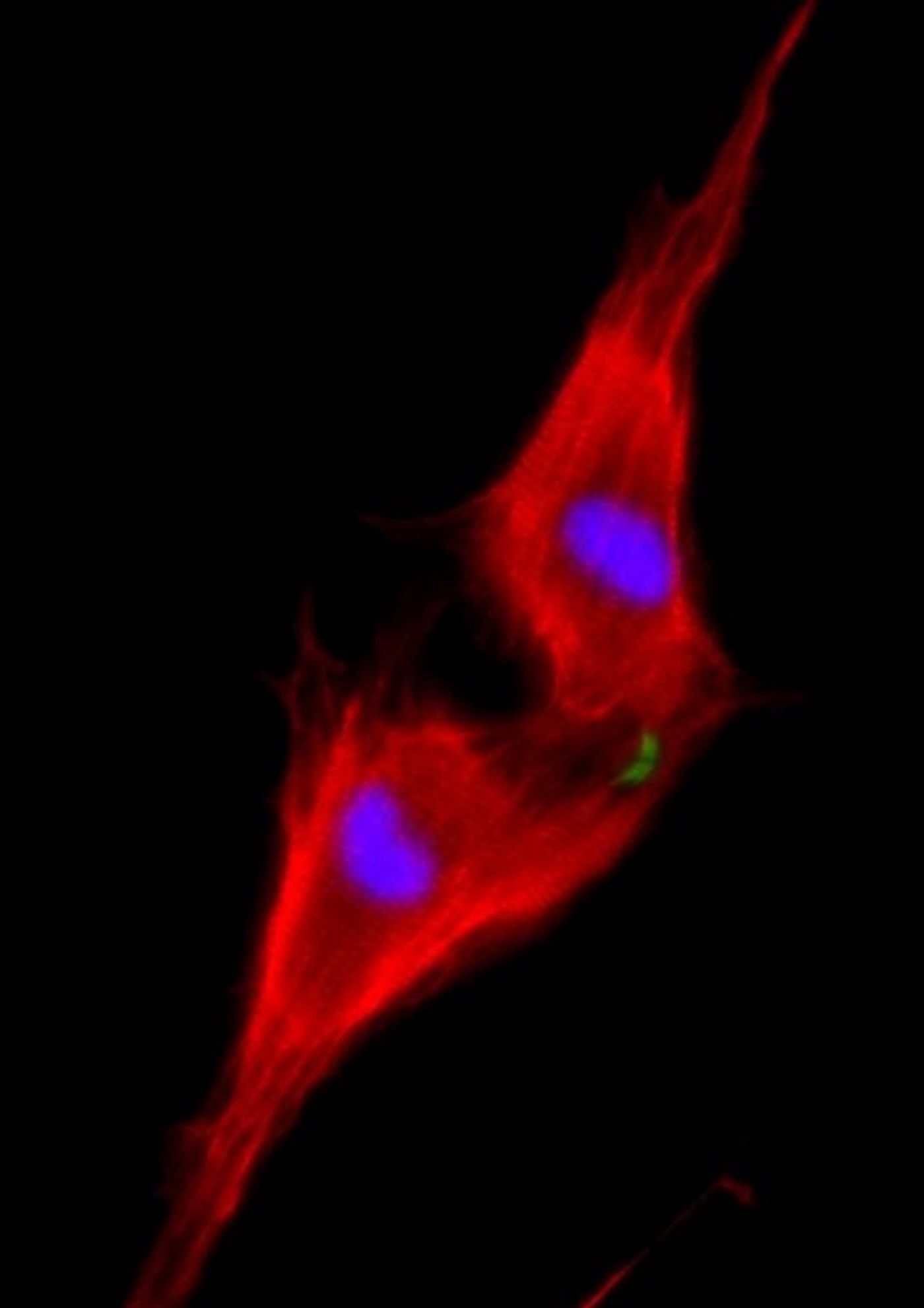
Professor Eldad Tzahor, PhD, of the Institute's Biological Regulation Department, thought part of the answer to the regeneration puzzle might lie in his area of expertise: embryonic development, especially of the heart. Indeed, it was known that a protein called ERBB2 - which is well studied because it can pass along growth signals promoting certain kinds of cancer - plays a role in heart development. ERBB2 is a specialized receptor - a protein that transmits external messages into the cell. ERBB2 generally works together with a second, related, receptor by binding a growth factor called Neuregulin 1 (NRG1) to transmit its message. NGR1 is already being tested in clinical studies for treating heart failure.
Gabriele D'Uva, PhD, postdoctoral fellow in Tzahor's research group, wanted to know exactly how NRG1 and ERBB2 are involved in heart regeneration. In mice, new heart muscle cells can be added up to a week after birth; newborn mice can regenerate damaged hearts, while seven-day-old mice already cannot. D'Uva and research student Alla Aharonov observed that heart muscle cells called cardiomyocytes that were treated with NRG1 continued to proliferate on the day of birth; but the effect dropped dramatically within a week, even with ample amounts of NRG1. Further investigation showed that the difference between a day and a week was in the amount of ERBB2 on the cardiomyocyte membranes.
The team then created mice in which the gene for ERBB2 was knocked out only in cardiomyocytes. This had a severe impact: The mice had hearts with walls that were thin and balloon-like - a cardiac pathology known as dilated cardiomyopathy. The conclusion was that cardiomyocytes lacking ERBB2 do not divide, even in the presence of NRG1. Next, the team reactivated the ERBB2 protein in adult mouse heart cells, in which cardiomyocytes normally no longer divide. This resulted in extreme cardiomyocyte proliferation and hypertrophy - excessive growth of the individual cardiomyocytes - leading to a giant heart (cardiomegaly) that left little room for blood to enter. "Too little or too much of this protein had a devastating impact on heart function," Tzahor says.
The question then became: If one could activate ERBB2 for just a short period in an adult heart following a heart attack, might it be possible to get the positive results (ie, cardiac cell renewal), without such negative ones as hypertrophy and scarring? Testing this idea, the team found that they could, indeed, activate ERBB2 in mice for a short interval only following an induced heart attack and obtain nearly complete heart regeneration within several weeks. "The results were amazing," Tzahor says. "As opposed to extensive scarring in the control hearts, the ERBB2-expressing hearts had completely returned to their previous state."
Investigation of the regenerative process through live imaging and molecular studies revealed how this happens: The cardiomyocytes "dedifferentiate" - that is, they revert to an earlier form, something between an embryonic and an adult cell, which can then divide and differentiate into new heart cells. In other words, the ERBB2 took the cells back a step to an earlier, embryonic form; and then stopping its activity promoted the regeneration process.
In continuing research, Tzahor and his team began to outline the pathway - the other proteins that respond to the NRG1 message inside the cell.
Tzahor says clinical trials of patients receiving the NRG1 treatment might not be overly successful if ERBB2 levels are not boosted as well. He and his team plan to continue researching this signaling pathway to suggest ways of improving the process, which may, in the future, point to ways of renewing heart cells.
This research, "ERBB2 triggers mammalian heart regeneration by promoting cardiomyocyte dedifferentiation and proliferation," appears in Nature Cell Biology.
[Source: Weizmann Institute of Science]