Stopping Cell Suicide
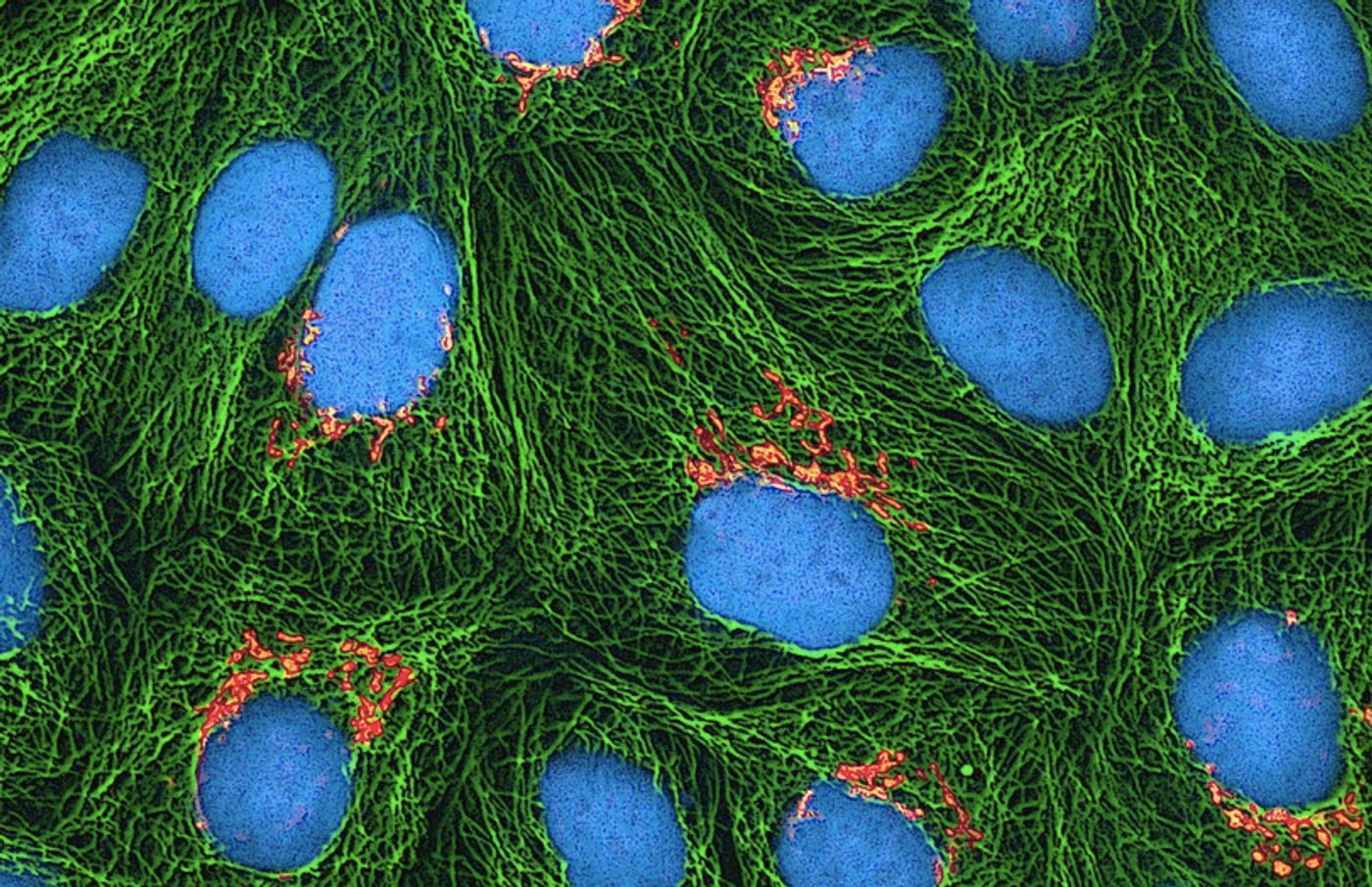
The body has a few ways to induce cell death when things are going awry, which is often an important process that maintains health. But sometimes, cell death is associated with diseases. Now researchers at Case Western Reserve University have learned more about this process, which may have significant implications for conditions in which cell death takes place, like arthritis and sepsis. When cells are undergoing excessive inflammation, one particular suicide pathway called pyroptosis is triggered; holes are ripped into cell membranes, leading to the death of the cell. Reporting in Science Immunology, researchers have found a way to stop that process.
"To date, there has been no pharmacologic mechanism to directly inhibit pyroptotic pore formation," noted study leader Derek Abbott, MD, Ph.D., Arline H. and Curtis F. Garvin Professor of Medicine at Case Western Reserve School of Medicine and member of the Case Comprehensive Cancer Center. "We knew necrosulfonamide inhibited another type of pore formation and had a hunch it might work against pyroptosis, too. We found necrosulfonamide is effective in our sepsis models and could be effective to treat diseases worsened by inflammation."
Inflammation is a common feature of many diseases, and pyroptosis can be a part of the destructive cycle that harms the body. When cells leak their contents out through holes in their membranes, the effect spreads. Molecules that leach out recruit more immune cells to the site, stimulating more pyroptosis. Chronic states of inflammation that can result are typical of disorders like IBD and arthritis. Abbott’s team sought a way to limit pyroptosis, without interfering with the essential duties of the immune system.
The looked to the known inhibitor of pore formation, necrosulfonamide, using it to treat mice that had been exposed to inflammatory bacteria. The researchers found that mice treated with the molecule survived longer than untreated mice.
When they applied the chemical to human cells growing in culture, they saw that even when inflammatory stimuli were used, immune cells remained unperturbed, and did not induce pyroptosis. Nothing leaked out of the cells, and they continued to function normally.
This research is the first test of necrosulfonamide as a pharmacologic agent, and more work will be required. But now we know it has potential as a therapeutic.
The team wanted to know more about how this molecule exerted its effect. They found that it binds to a molecule called gasdermin D, which can destroy cell membranes. When pyroptosis happens, gasdermin D builds up in long chains inside of the cell membrane, creating holes.
Necrosulfonamide physically prevents that buildup from happening. It's a big molecule that attaches to gasdermin D where it would otherwise attach to other gasdermin D molecules. With necrosulfonamide attached to it, it cannot aggregate. That process is different from preventing cell death chemically.
"Our results suggest gasdermin D inhibitors are clinically possible," added first author Joseph Rathkey, a student in the MSTP at Case Western Reserve School of Medicine. "We've provided proof of principle that destructive pore formation can be inhibited, and that gasdermin D is a viable pharmaceutical target." This work may also inform the design of other drugs.
Learn more about different kinds of cell death used by the body from the video.
Sources: AAAS/Eurekalert! Via Case Western Reserve University, Nature Reviews Microbiology, Science Immunology