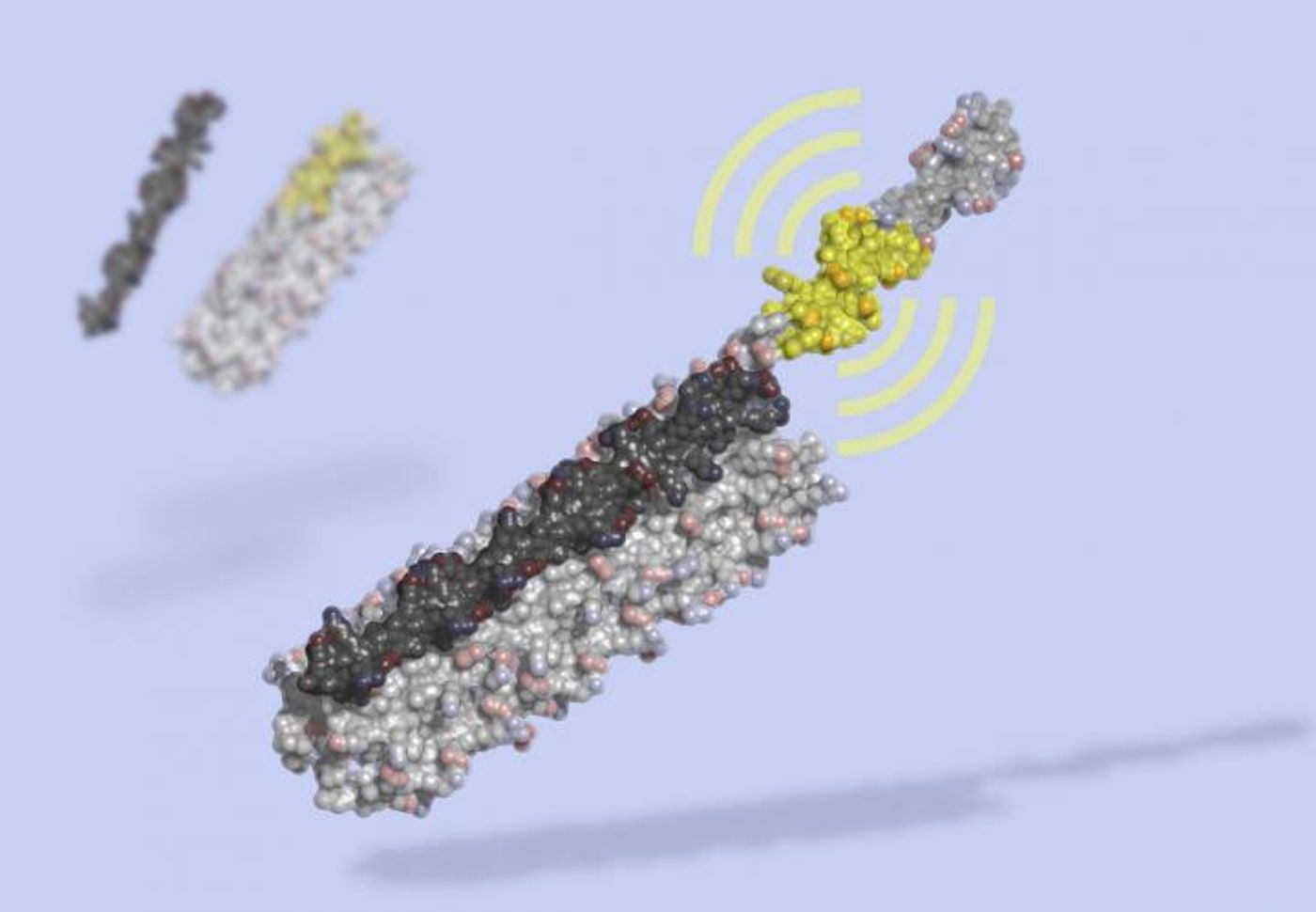
LOCKR: A New Way to Control Cellular Function Synthetically
A totally synthetic system has been created to act like a switch that can change gene expression, protein interactions, and the biological functions in a cell. The synthetic protein 'switch' is called LOCKR (Latching, Orthogonal Cage/Key pRotein); it's been described and demonstrated at work in two papers in Nature by the research teams of David Baker at the UW Medicine Institute for Protein Design and Hana El-Samad at UC San Francisco. The technology is outlined in the video.
"The ability to control cells with designer proteins ushers in a new era of biology," said El-Samad, the Kuo Family Professor of Biochemistry and Biophysics at the University of California San Francisco and co-senior author of the reports. "In the same way that integrated circuits enabled the explosion of the computer chip industry, these versatile and dynamic biological switches could soon unlock precise control over the behavior of living cells and, ultimately, our health."
There is no natural analog for LOCKR, which is different from CRISPR, which uses and enzyme and a guide RNA molecule to make cuts in the genome. LOCKR can be used in various ways, for example, to modify gene expression, change trafficking in the cell, trigger the degradation of specific proteins, and control how some proteins interact. It can be utilized to generate biological circuits that can act as sensors, detecting environmental conditions, and responding in certain ways.
"Right now, every cell is responding to its environment," explained a lead author of one study, Bobby Langan of the University of Washington Medicine Institute for Protein Design. "Cells receive stimuli, then have to figure out what to do about it. They use natural systems to tune gene expression or degrade proteins, for example."
LOCKR is only eight nanometers wide on its longest side when it's assembled - over one hundred million of them would be needed to span the period ending this sentence.
In this work, computational tools were used to design proteins that are triggered to self-assemble into bioactive peptides once specific molecular keys were present.
Once the team installed this LOCKR system in a yeast model, they showed that the bioengineered organism could break down a specific protein exactly when the researchers wanted it done. A different version of the switch initiated the same process in human cells grown in the lab.
LOCKR may offer a new therapeutic avenue for diseases that involve aberrant gene expression or protein activity. It is an entirely new way for researchers to tinker with cellular function.
"LOCKR opens a whole new realm of possibility for programming cells," said a lead study author Andrew Ng of the UC Berkeley-UCSF Graduate Program in Bioengineering. "We are now limited more by our imagination and creativity rather than the proteins that nature has evolved."
Sources: AAAS/Eurekalert! via UW Medicine, Nature