RNA Polymerases Can Signal to One Another Over Long Genomic Distances
Each cell of an organism carries its genome in the nucleus and the genes that are expressed in that cell help establish its identity and ensure it functions correctly. Active genes are transcribed by cellular machinery into a molecule called RNA (animated in the video below). Protein-coding genes then get translated, and further processed by the cell before going on to serve their purpose. Scientists have learned a lot about transcription by studying large groups of cells that grow in the tissues of an animal, or in laboratory cultures. But research tools are rapidly advancing, and now scientists are able to examine the processes used by individual cells, like transcription, more closely.
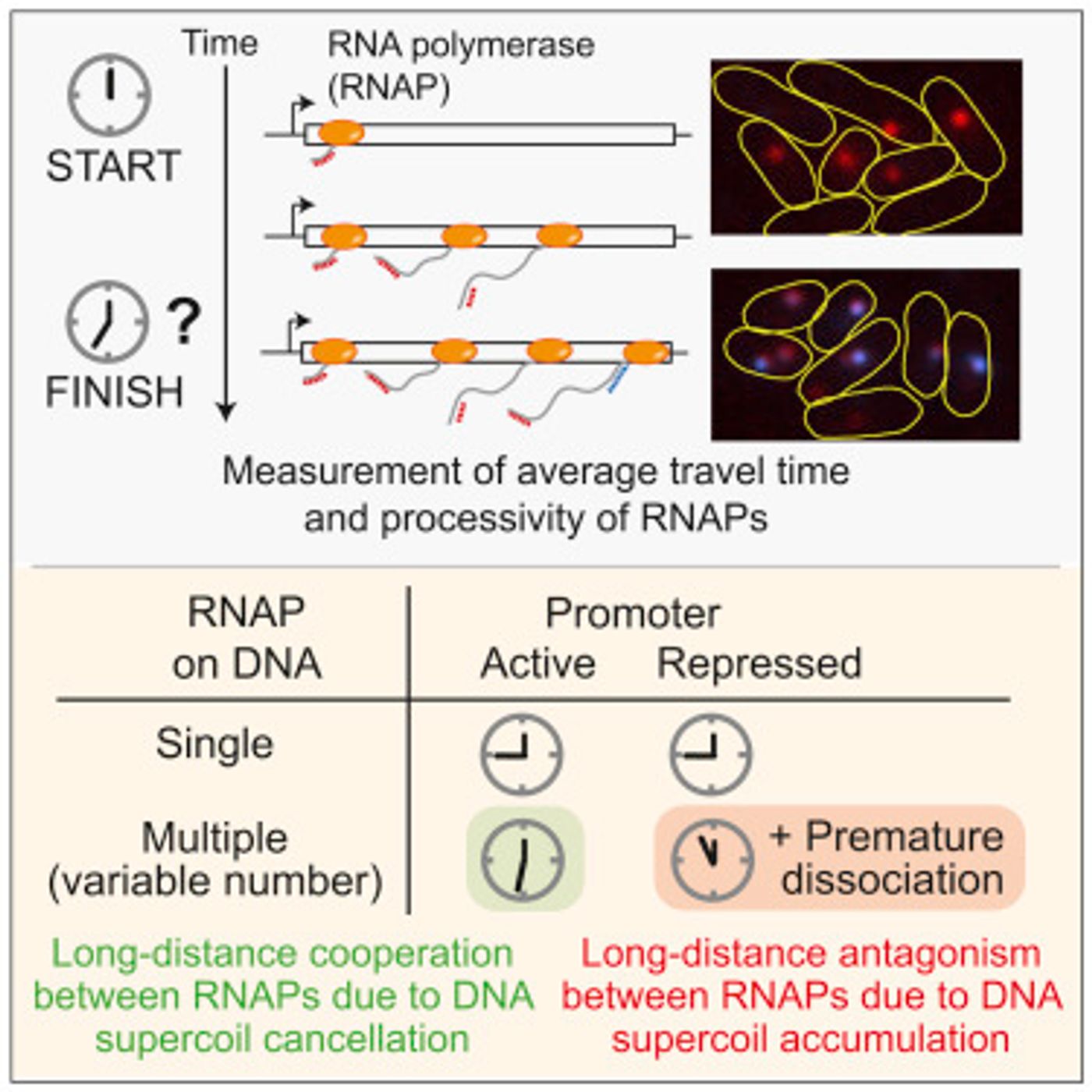
Scientists at the University of Illinois at Urbana-Champaign have now taken a close look at transcription in the Escherichia coli bacterium at the level of a single molecule. A study reported in Cell and led by the team of Professor Sangjin Kim has indicated that long-distance communications between the enzymes that transcribe DNA exist and impact transcription dynamics. These enzymes are called RNA polymerases.
In this study, the research team collaborated with scientists at Yale University’s Microbial Sciences Institute to assess transcription in vitro and in vivo while multiple RNA polymerases (RNAPs) worked on the gene at the same time. These polymerases sit at different points on the gene, and the researchers observed interactions, which sped or slowed transcription depending on the cell’s environment. Even when the RNA polymerases were separated by two thousand nucleotide bases, they could still signal to one another.
"This communication is coming from a property of DNA called supercoiling, which dynamically changes during both replication and transcription. DNA is a double helix that supercoils—like a rope that can be twisted—in response to a portion of the double-strand being opened so that it can be read," explained Kim. "The twisting is a natural consequence of the function. In transcription, as RNAP translocates along the DNA, DNA becomes twisted, but our study shows that this, in turn, becomes a mechanism allowing for the long-distance communication between RNAPs."
Genes are coupled with promotors that can turn transcription on and off in response to the needs of the cell. The researchers also determined that when the promotor was ‘off,’ the RNA polymerases slowed down and disengaged from the gene.
This work may help shed light on how mutations are introduced to DNA or other molecular mechanisms. "It is known that when RNAP stops, it can introduce mutations on the genetic code. This has possible implications for the rise of antibiotic resistance in bacteria. In future work at the CPLC [Center for the Physics of Living Cells], I want to test this by modulating DNA supercoiling and looking deeper at the mechanics. This line of inquiry provides a great opportunity for collaboration between theory and experiment," added Kim.
"My dream project is to probe the physical mechanism of this emergent phenomenon—the coupling between DNA supercoiling and RNA polymerase motion. The CPLC is a fantastic place to pursue this and to collaborate with other theoretical and experimental scientists at the intersection of microbiology, biochemistry, and biophysics," noted Kim. "I have already connected with wonderful new colleagues who are interested in collaborating on this, including Ido Golding, Nigel Goldenfeld, and Yann Chemla."
Learn more about the CPLC from the video.
Sources: Phys.org via University of Illinois, Cell