Scientists Create a New Version of CRISPR - Prime Editing
Scientists at the Broad Institute of MIT and Harvard have taken step toward improving genetic engineering by creating a new form of CRISPR. Their new technique is called ‘prime editing,’ and it can make more precise and efficient edits to the genome. The researchers suggested that it might be able to repair up to 89 percent of genetic errors that cause disease. The work has been reported in Nature.
"A major aspiration in the molecular life sciences is the ability to precisely make any change to the genome in any location. We think prime editing brings us closer to that goal," said the senior author of the report David Liu, who is Richard Merkin Professor, vice-chair of the faculty, and director of the Merkin Institute of Transformative Technologies in Healthcare at the Broad Institute of MIT and Harvard. "We're not aware of another editing technology in mammalian cells that offers this level of versatility and precision with so few byproducts."
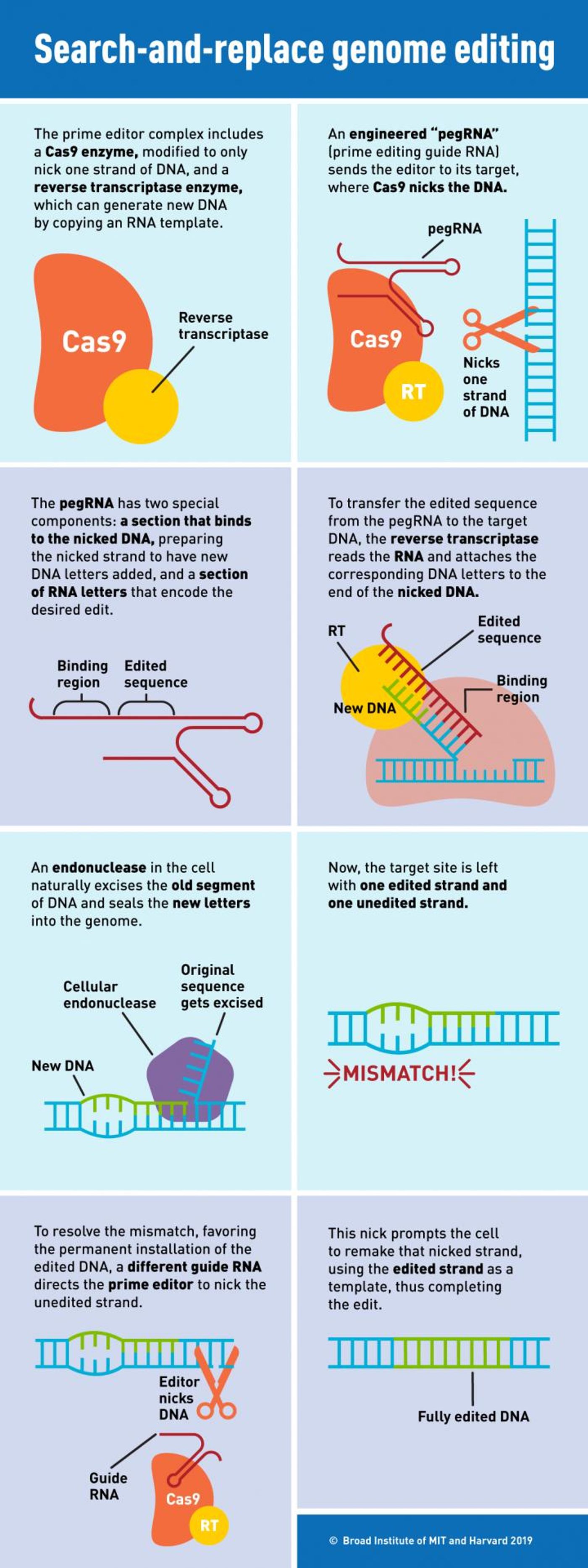
The CRISPR/Cas9 system makes edits to the genome by cutting strands of DNA. Certain variations on the method can cut only one strand and leave the other intact, and the tool can also be combined with a DNA repair template for correcting mutations in the genome. Prime editing instead uses RNA to insert new genetic sequences into the genome of human cells. Previous work by Liu’s team has created base editors, which change a single base pair of the genome to a different nucleotide. Currently, base editors are able to change Cytosine (C) to Thymine (T), Adenine (A) to Guanine (G), and the reverse of those reactions: T to C or G to A.
Liu discusses the repair of single base-pair mutations in the video.
Now they have combined prime editing with an enzyme called reverse transcriptase. This enzyme can generate DNA from an RNA sequence, and ‘primes’ a target area in the genome for the insertion of new DNA. This new method employs a molecule called pegRNA, which directs the editor to the targeted portion of the genome, and also carries the new sequence that’s to be inserted. Reverse transcriptase then uses the molecule to edit in the new nucleotides in the proper location.
In their report, the researchers demonstrated the method by correcting genetic mutations that cause sickle-cell anemia. In one very specific base out of billions, a T was changed to A. They were also able to remove four nucleotides that are to blame for Tay Sachs disease as well as perform several other successful edits in human and mouse neurons. They efficiently changed one base to another in all of the twelve possible combinations of the swaps, were able to precisely remove up to 80 nucleotides, and insert as many as 44 new ones.
"With prime editing, we can now directly correct the sickle-cell anemia mutation back to the normal sequence and remove the four extra DNA bases that cause Tay Sachs disease, without cutting DNA entirely or needing DNA templates," noted Liu, who is also a Howard Hughes Medical Institute investigator. "The beauty of this system is that there are few restrictions on the edited sequence. Since the added nucleotides are specified by the pegRNA, they can be sequences that differ from the original strand by only one letter, that have additional or fewer letters, or that are various combinations of these changes."
"The versatility of prime editing quickly became apparent as we developed this technology," said postdoctoral fellow and the study’s first author Andrew Anzalone. "The fact that we could directly copy new genetic information into a target site was a revelation. We were really excited."
The scientists found that few undesired or off-target edits were made with their new tool compared to CRISPR methods that cut both strands of the genome. It’s also better at making changes to individual nucleotides than typical methods when applied to areas of the genome that can be difficult to modify.
The researchers are planning to refine and improve prime editing further; they want to make it even more efficient as well as investigate the consequences of these edits. They are interested in eventually using this technology to help people with genetic diseases.
Sources: AAAS/Eurekalert! via Broad Institute of MIT and Harvard, Nature