New CRISPR-HOT Technique Can Color Cells and Genes
Since the CRISPR/Cas9 gene-editing tool was developed several years ago, many scientists have modified and improved it for different applications. It consists of a molecule that can target a particular location on a genome, and an enzyme that in the case of Cas9, cuts the genomic DNA at that targeted location. Scientists have now used the concept to fluorescently label genes and cells in simplified, miniature versions of organs that are grown in culture, called organoids.
Reporting in Nature Cell Biology, the scientists applied a CRISPR method that doesn't only cut the genome, it can also incorporate new DNA into the genome where the cut is made. While that has been possible with CRISPR before, in this work, the researchers have used an alternative methodology that they've called CRISPR-HOT. They've applied it to the study of liver cells called hepatocytes. They wanted to know more about how these cells divide and the appearance of abnormal cells that carry too much DNA.
When Cas9 is guided to the genome and it makes a cut, the cell works to fix it. "The little wound in the DNA can activate two different mechanisms of repair in the cells, that can both be used by researchers to coerce the cells to take up a new part of DNA, at the place of the wound," explained Delilah Hendriks of the Hubrecht Institute.
"Since some earlier work in mice indicated that new pieces of DNA can be inserted via non-homologous end joining (NHEJ), we set out to test this in human organoids," said Benedetta Artegiani of the Hubrecht Institute. It had been thought that NHEJ was error-prone, and wasn't used often. This work demonstrated that inserting DNA into human organoid cells was more efficient using NHEJ, however. The scientists called the technique CRISPR-HOT.
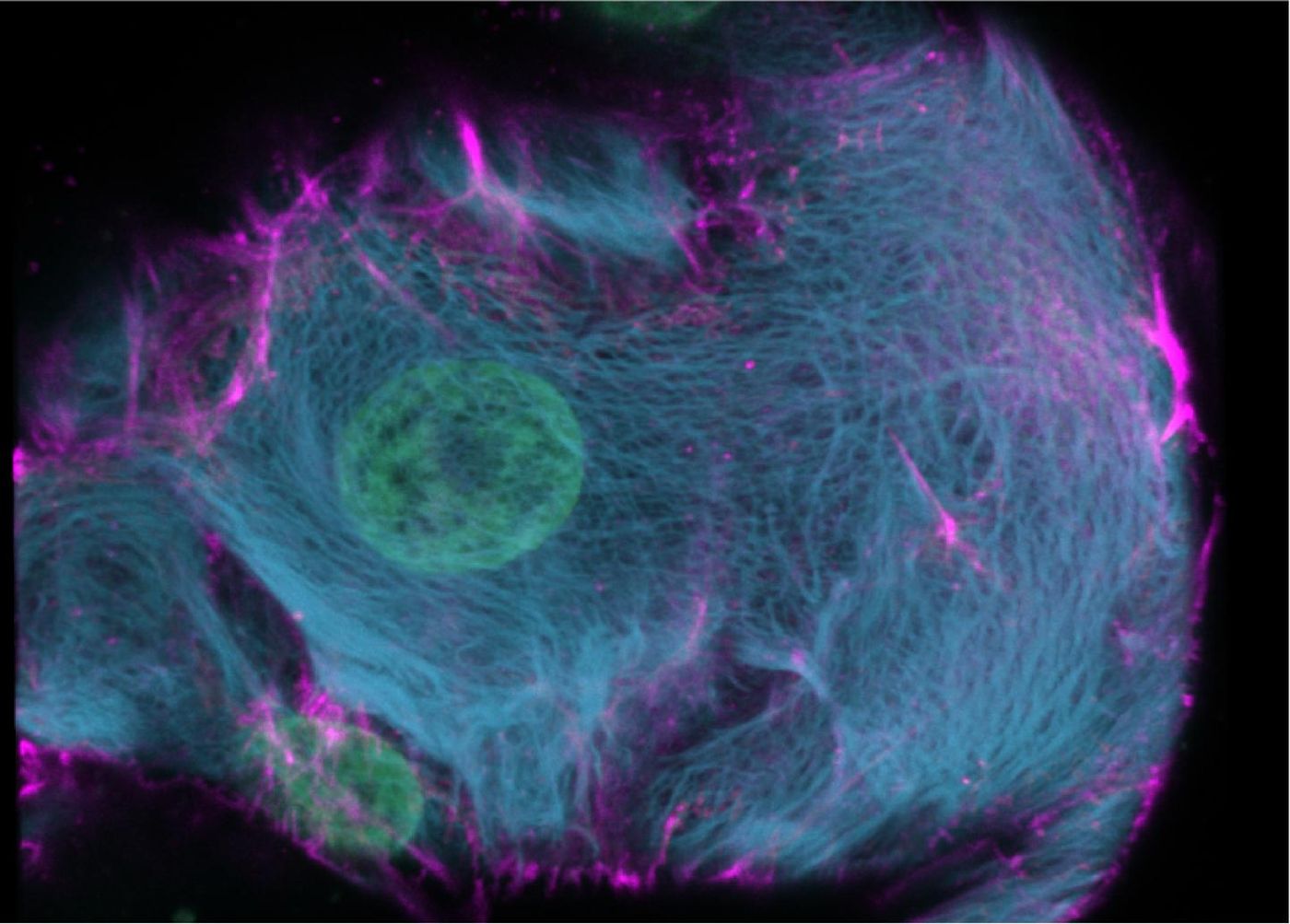
The researchers used CRISPR-HOT to add fluorescent tags to specific genes of interest in the DNA of human organoids. They labeled rare, hormone-producing intestinal cells called enteroendocrine cells, making them easy to identify. They also labeled organoids that were generated from a liver cell type called biliary ductal cells. CRISPR-HOT enabled the scientists to observe the ultrastructure of keratin proteins, which change as cells specialize.
Many liver cells carry two or meter times the normal amount of DNA, but it's not known how they form or whether they divide. There are more of these cells in older adults, but it's unclear if they're linked to disease. Artegiani and Hendriks were able to study cell division in hepatocytes using CRISPR-HOT.
"We saw that "normal" hepatocytes divide very orderly, always splitting into two daughter cells in a certain direction," said Artegiani.
"We also found several divisions in which an abnormal hepatocyte was formed. For the first time we saw how a "normal" hepatocyte turns into an abnormal one," said Hendriks.
The researchers also examined the impact of a mutation in a gene called TP53, which can cause abnormal cell division in hepatocytes. Without the functional gene, the hepatocytes began to divide more often, which may lead to cancer development.
This tool may be especially useful in the study of cell differentiation and development, as well as abnormal cells.
Sources: AAAS/Eurekalert! via Hubrecht Institute, Nature Cell Biology