A Potential Breakthrough in Type 1 Diabetes Treatment
After a meal, our body has to regulate the amount of sugar in the bloodstream and does so through a molecule called insulin, which helps move sugar to the liver, muscles, and fat. Insulin is made by cells in the pancreas called beta cells. In type 1 diabetes, these cells are erroneously targeted for destruction by the immune system, and the body no longer produces insulin. Type 1 diabetes is a genetic disorder, and can be caused by mutations in several different genes. Patients with her disease have to manage the condition for their whole lives.
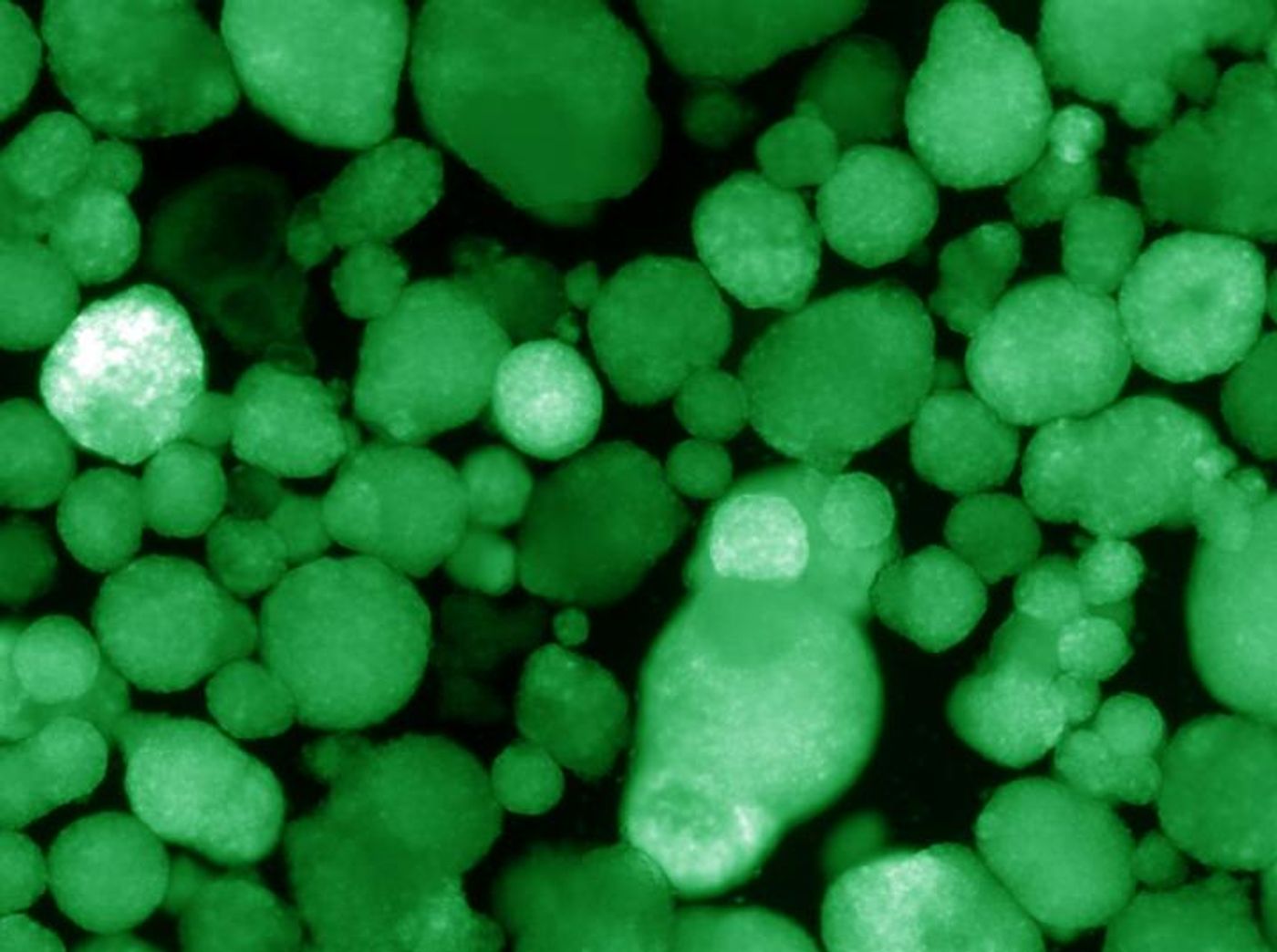
Reporting in Nature, researchers have now made a major breakthrough in the development of a better treatment that goes beyond monitoring glucose levels and injecting insulin. They used stem cells to engineer the first human pancreatic cell clusters that produce insulin and escape the damage that the immune system inflicts on these cells in type 1 diabetes. The clusters were able to control glucose levels in the blood after they were transplanted in mice, without the use of immunosuppressants.
"Most type 1 diabetics are children and teenagers," said Salk Professor Ronald Evans, senior study author and holder of the March of Dimes Chair in Molecular and Developmental Biology. "This is a disease that is historically hard to manage with drugs. We hope that regenerative medicine in combination with immune shielding can make a real difference in the field by replacing damaged cells with lab-generated human islet-like cell clusters that produce normal amounts of insulin on demand."
Treatment options for type 1 diabetes have emerged over time. It is possible to transplant clusters of beta cells - pancreatic beta islets from healthy donors into type 1 diabetes patients, but these individuals must then take drugs that suppress the immune system and come with their own risks. Insulin pumps can make the process of getting insulin a little easier, but the condition still has to be carefully managed. This work may be great news for the estimated 1.6 million Americans that have type 1 diabetes.
Led by Evans, researchers were able to use stem cells to generate beta-like cells that made insulin. However, they did not release that insulin in response to glucose. Evans explained that the cells simply needed a boost, which could be provided by altering a gene called ERR-gamma.
"When we add ERR-gamma, the cells have the energy they need to do their job," said study co-author Michael Downes, a Salk senior staff scientist. "These cells are healthy and robust and can deliver insulin when they sense high glucose levels."
The cells were grown in a three-dimensional culture system that models the pancreas more closely than a dish of cells. They also determined that a protein called WNT4 can activate the power switch triggered by ERR-gamma. After using this data to create human islet-like organoids (HILOs), they moved to immure rejection. They found that a protein called PD-L1 can shield the transplanted cells.
"By expressing PD-L1, which acts as an immune blocker, the transplanted organoids are able to hide from the immune system," said the first study author Eiji Yoshihara, a former staff scientist in the lab.
After stimulating PD-L1 in HILOs with bursts of another protein, interferon gamma, the transplants worked to control blood glucose in a diabetic mouse model. More work will be needed before the strategy moved to clinical trials, but the researchers are hopeful.
"This is the first study to show that you can protect HILOs from the immune system without genetic manipulation," said Downes. "If we are able to develop this as a therapy, patients will not need to take immune-suppressing drugs."
Sources: AAAS/Eurekalert! via Salk Institute, Nature