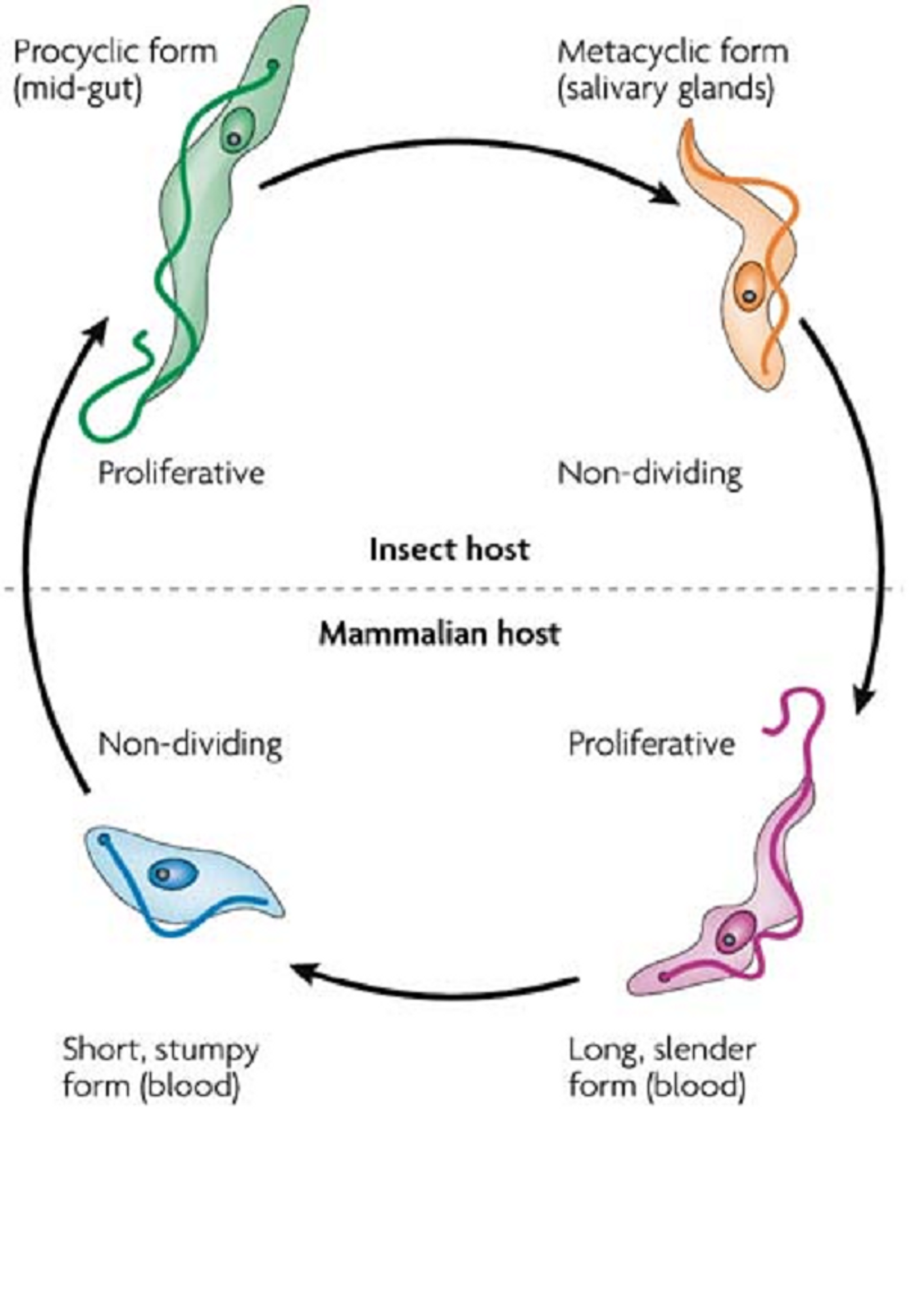
Trypanosoma brucei is a complex of protozoan parasites that cause a disease known as African sleeping sickness. There are two subspecies that are morphologically indistinguishable from each other:
T.b. gambiense (West African sleeping sickness) and
T.b. rhodesiense (East African sleeping sickness). The life cycle of the parasite starts with the tsetse fly, which injects the parasites into the skin tissue of a mammalian host during a blood meal. The parasites then enter into the bloodstream through the lymphatic system and are carried to other parts of the body where they continue to replicate. Another tsetse fly feeds on the same mammalian host and becomes infected. The parasite goes through a variety of extracellular stages in the midgut of the tsetse fly and continues to replicate in the fly’s salivary glands. The entire life cycle of
Trypanosoma brucei takes about 3 weeks.
Depending on whether the infection is caused by subspecies
T.b. gambiense or subspecies
T.b. rhodesiense will determine the type of clinical disease that appears in humans. Common symptoms include fever, headache, muscle joint aches, and malaise. Either form will eventually lead to coma and death if not appropriately treated.
In order to survive and complete their life cycle, parasites must be able to switch genes on and off depending on whether cells want to divide or differentiate. The differentiation of
T. brucei occurrs within the mammalian host, when the growth stage is halted, to the replicative forms within the midgut of the tsetse fly. This differentiation was found to be induced by cis-aconitate (CA). Cis-aconitate intermediates the isomerization of citrate to isocitrate during the citric acid cycle.
In a study performed by researchers at the University of Edinburgh, scientists found that
T. brucei cells only responded to CA when exposed to a threshold of over 1 mM CA however; after exposure to CA for 2-3 hours, the cells finally committed to differentiation from the “stumpy” (non-growth) stage within the mammalian most to the “procyclic” (growth) stage within the insect vector. The cells continued to be committed to this differentiation even if CA was depleted from the environment.
These results led researchers to conclude that the transition between cell stages of
T. brucei is mediated by an irreversible bistable switch. Bistable switches are commonly used to govern biological events within cells, such as during mitosis. They believe that this switch enables the parasites to be quick and decisive in terms of their commitment to differentiation during environmental change. In addition, researchers found that protein synthesis was required for
T. brucei to “remember” the signal which induced cell differentiation. After 3 hours exposure to CA, there were numerous changes observed within the parasites proteome, specifically related to changes in protein phosphorylation and expression of protein kinase (NRK). This research provides insight into the fundamental life cycles of human parasitic pathogens including how these parasites progress to the next steps within the life cycle. This decision allows the parasite to avoid death in a changing environment.
Sources:
Journal of Cell Biology;
Centers for Disease Control and Prevention