Bringing Light to Extracellular Vesicles
Written By Christopher Pratt, PhD
Origin of extracellular vesicles
Though originally thought of as strictly a waste disposal system, extracellular vesicles (EVs) have been shown to have myriad cell communication functions. Recent research has resulted in a deeper understanding of the role these vesicles play and is leading to innovation in diagnostics and therapeutics in fields like neurodegenerative disease, cancer biology, and immunology.
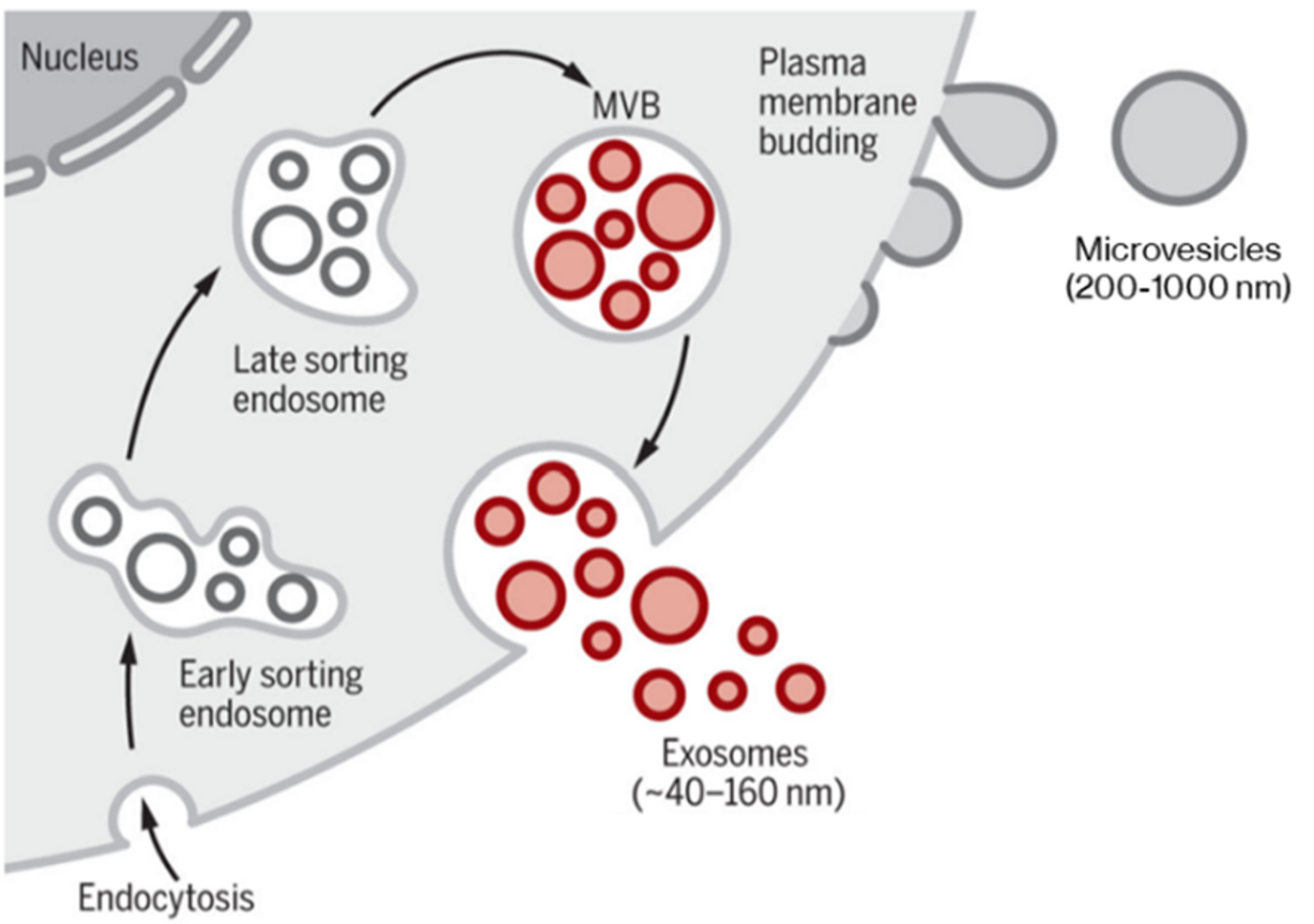
Two main classes of EVs exist: microvesicles are large (200-2000 nm diameter) vesicles produced by direct budding from the plasma membrane, while exosomes are small (40-200 nm) and primarily produced when a multivesicular body fuses with the plasma membrane, releasing intraluminal vesicles as exosomes. Exosomes directly budding from the plasma membrane have been observed in T-cells; however, the frequency of this phenomenon in other cell types is unclear.1
Classical modes of EV generation, producing microvesicles directly from plasma membrane and exosomes via the late sorting endosome and multivesicular body (MVB). Certain cell types can also produce exosomes by direct plasma membrane budding. Adapted from Kalluri & LeBleu (2020).2 Reproduced under the Creative Commons license.
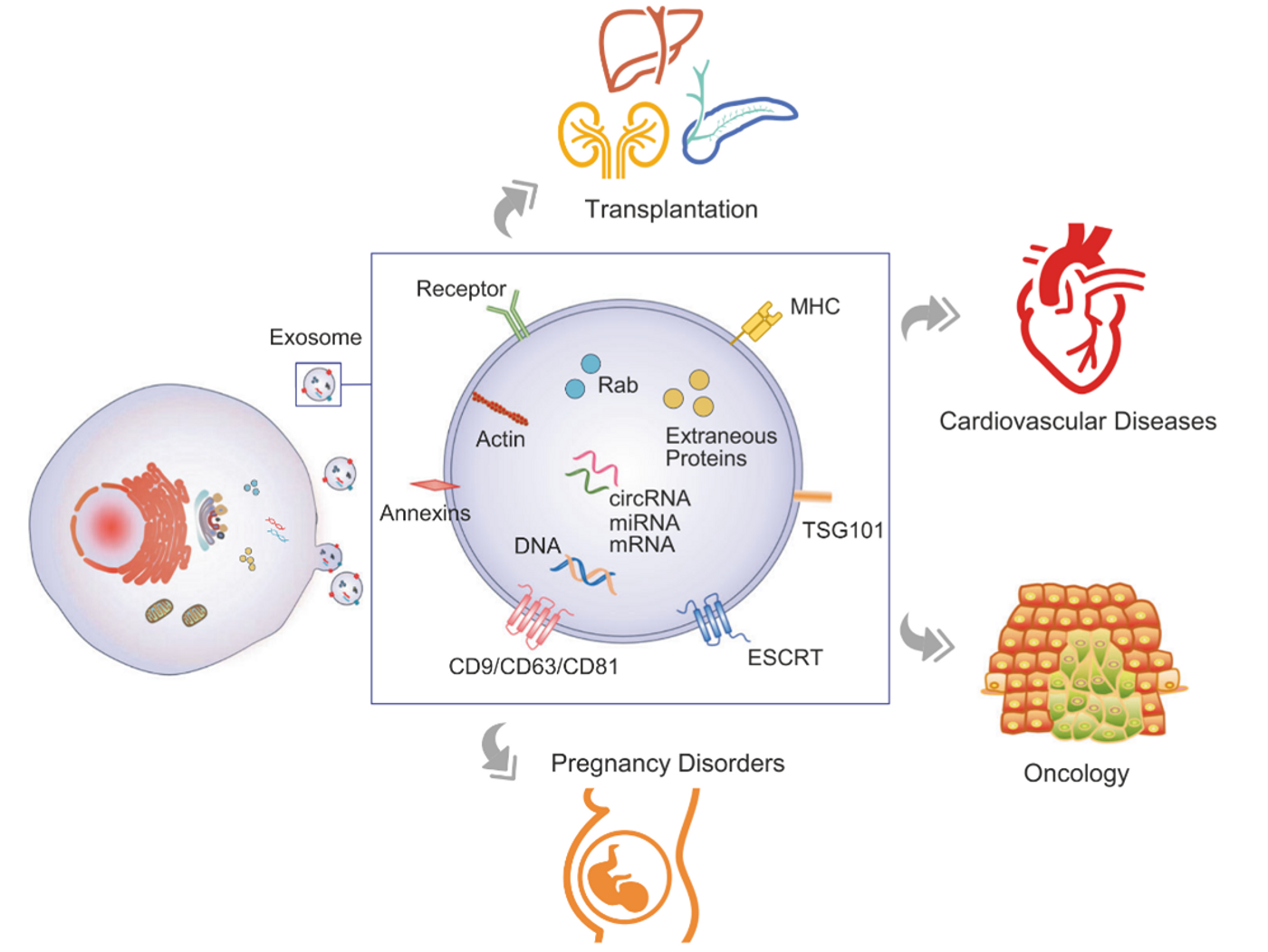
EVs have proteomes that resemble the originating cell but are enriched in certain proteins, such as the tetraspanins—membrane-bound molecular organizers—CD9 and CD81 in microvesicles and exosomes, and CD63 specifically in exosomes. Exosomes contain a dense intraluminal core of scaffolding and aggregated protein bound with chaperones, primarily HSP70, and can also carry nucleic acids.
Extracellular vesicles in disease and diagnostics
EVs carry payloads of proteins, RNA, and DNA that function in recipient cells.2 Microvesicles secreted by cancer cells (occasionally termed oncosomes) can transport oncogenic mRNA that promotes invasion and metastasis in breast cancer cells.3 EVs can also contribute to immunity and immunosuppression: antigen presenting cells secrete exosomes containing major histocompatibility complex class II bound with antigen, which can promote adaptive immunity over a distance.2
Exosomes have also been associated with some neurodegenerative conditions. For example, prion protein is present in brain-derived exosomes, where it affects immunomodulation, plasticity, and normal brain function. Exosomes have been observed to carry pathogenic prion protein and potentiate its neurotoxic effect in spongiform encephalopathies.4 MicroRNAs and proteins associated with disease progression of Alzheimer’s and Parkinson’s have been found in brain-derived exosomes.4,5
Liquid Biopsies
One of the most exciting prospects for exosome research is developing non-invasive “liquid biopsies”—using blood samples to monitor for disease or to sample inaccessible organs such as the brain or pancreas.6 Liquid biopsies could be used for early detection of cancers, neurodegenerative disease, pregnancy disorders, and much more.6
Circulating EVs make non-invasive liquid biopsies possible. Figure from Zhou et al. 2020.6 Reproduced under the Creative Commons license.
The challenges of analyzing EVs
As small heterogeneous extracellular particles, characterizing EVs is challenging; one barrier is a lack of standardized isolation and analysis methods.2 EVs have classically been isolated by ultracentrifugation or sucrose gradient ultracentrifugation, but the purity of these isolated EVs is poor. New isolation techniques such as acoustic nanofiltering and microfluidic immuno-isolation improve purity but require specialized equipment.
Scanning and transmission electron microscopy can readily visualize EVs, but sample preparation creates artifacts and can introduce sampling bias.7 Fluorescence imaging with lipophilic dyes in culture systems is confounded by other small particles in biofluids and cell culture media like serum proteins and lipoproteins.8
Analyzing exosomes with flow cytometry is a new innovation, although it is challenging to distinguish exosomes from cellular debris. It is also crucial to avoid forming aggregates, as the loss of individual resolution can be disastrous for heterogeneous populations. Fortunately, EVs have well-defined protein markers that can be labeled such as the tetraspanins.1,7 EV flow cytometry validated antibodies are a key starting point and membrane stains validated for flow cytometry are useful for measuring EV size by fluorescence intensity.
The Future of EVs
EVs represent an exciting and booming field of cell biology with life-changing diagnostic and therapeutic potential. Better understanding and characterization of EVs is critical in making this a reality. If you are looking to study exosomes in your research, check out Biotium’s curated list of validated antibodies and probes for EV detection. Without a doubt this field will continue to drive advances in cell biology and medicine as more efficient tools are developed to study EVs.
References
- van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol 19, 213–228 (2018).
- Kalluri, R. & LeBleu, V. S. The biology, function, and biomedical applications of exosomes. Science 367, (2020).
- Le, M. T. N. et al. miR-200-containing extracellular vesicles promote breast cancer cell metastasis. J Clin Invest 124, 5109–5128 (2014).
- Song, Z. et al. Brain Derived Exosomes Are a Double-Edged Sword in Alzheimer’s Disease. Front. Mol. Neurosci. 13, (2020).
- Yu, H. et al. Potential Roles of Exosomes in Parkinson’s Disease: From Pathogenesis, Diagnosis, and Treatment to Prognosis. Front. Cell Dev. Biol. 8, (2020).
- Zhou, B. et al. Application of exosomes as liquid biopsy in clinical diagnosis. Signal Transduction and Targeted Therapy 5, 1–14 (2020).
- Shao, H. et al. New Technologies for Analysis of Extracellular Vesicles. Chem Rev 118, 1917–1950 (2018).
- Takov, K., Yellon, D. M. & Davidson, S. M. Confounding factors in vesicle uptake studies using fluorescent lipophilic membrane dyes. J Extracell Vesicles 6, (2017).