Silent Genetic Substitutions Can Affect Protein Function
It was long thought that unless a mutation in a gene's sequence changed the resulting amino acid, it had no significance - the mutation was said to be 'silent.' But researchers have been questioning that dogma. We know that proteins are made of amino acids, and that a gene's sequence determines which amino acids are used to make a protein; a codon of three nucleotide bases encodes for one amino acid. There is also redundancy in the genetic code; in most cases, several combinations of amino acids encode for one amino acid (tryptophan is the only amino acid specified by only one codon).
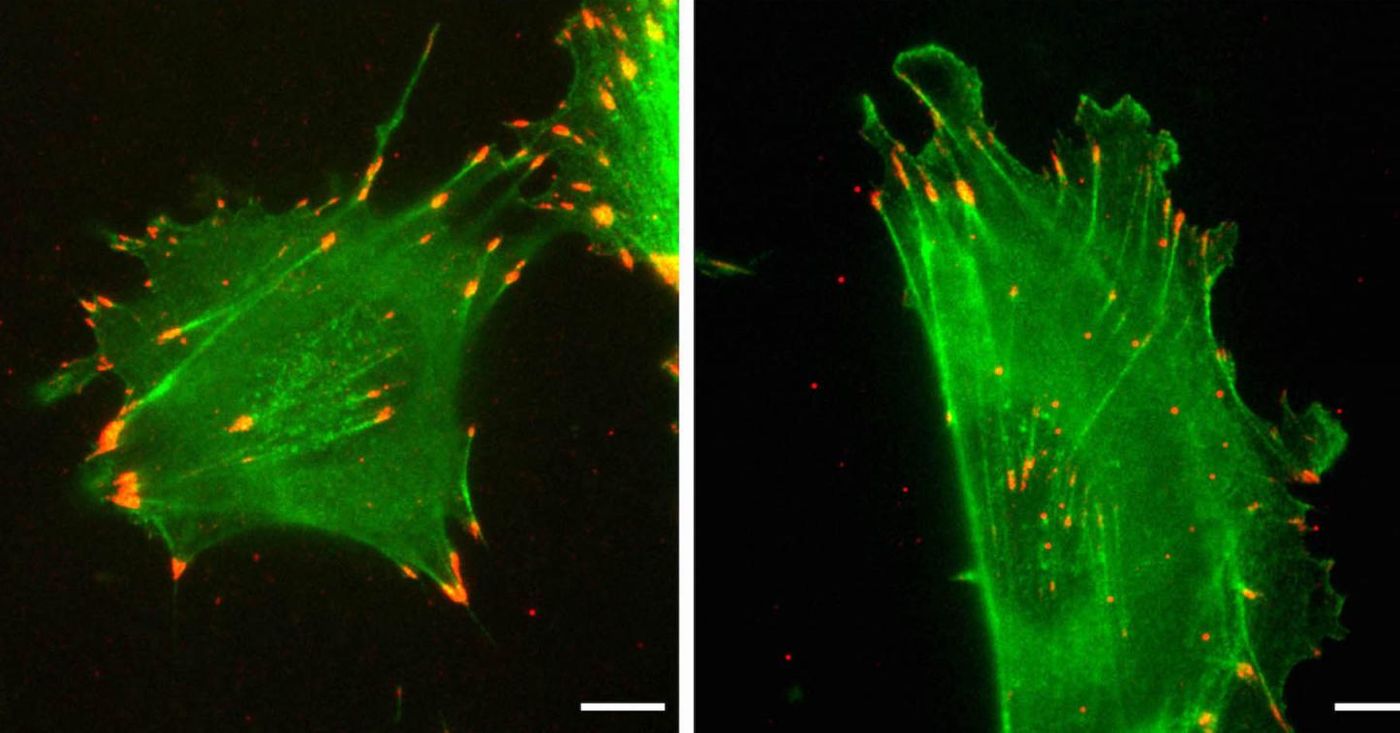
Scientists have been studying silent mutations in a protein called actin. There are a few types of actine protein, in this work the researchers focused on beta-actin and gamma-actin. The amino acids that make up these proteins are 99 percent similar so they're almost the same, but they have different functions. Reporting in eLife, researchers have found that the 13 percent of the sequences of these actin genes that are 'silently' different are causing the proteins to take on very different roles in the cell.
"On a global, philosophical level, this expands our understanding of genetic code," said senior study author Anna Kashina, a professor of biochemistry at Penn University's School of Veterinary Medicine. "We used to believe that the role of the nucleotides was to encode amino acids, but now we see that, actually, proteins with the same amino acid sequence have different translation rates, and that makes a difference in their function."
The study found that the mRNA molecules that encode for beta-actin get translated into protein more quickly than gamma-actin mRNA does. Both proteins are involved in cell movement, but beta-actin appears to slow a cell down by causing it's surface attachment to get stronger.
Previous work by the Kashina lab has indicated that silent genetic edits in mice could cause their gamma-actin to act like beta-actin. The researchers had also observed that ribosomes, which generate protein from mRNA, were more densely packed on beta-actin RNA compared to gamma-actin RNA; they hypothesized that this could explain the different rates of protein synthesis and subsequent changes in functional roles.
"We expected that faster translation would mean faster movement, and that's not what we found. It took us a long time to explain why," said Kashina.
In a cell culture model of wounds, differences in the nucleotide sequence of the actins had an effect on the rate of healing. Cells that only expressed beta-actin migrated and healed a tear normally, but cells that only expressed gamma-actin moved twice as quickly. When edits were made that swapped the actin sequences, the rates of cell movement followed the sequence.
When beta-actin was given the amino acid sequence of gamma-actin in edited cells, they moved like cells that express gamma-actin. If gamma-actin was given beta-actin's amino acid sequence, the cells moved like beta-actin-expressing cells.
Although some beta-actin was generated faster than gamma-actin, that doesn't aid the speed of cell migration. "We found that the faster you supply it, the better the cell attaches to the substrate. It creates proper traction, which is essential for normal migration. And if you don't supply it fast enough the cell can't attach properly and starts sliding. So that explained our seemingly counterintuitive results," noted Kashina.
"We think this is part of a bigger story," Kashina said. "We believe actins are not the only proteins that behave this way. There are a number of protein families in the human genome that contain highly similar proteins encoded by different genes. This silent code could be at play in those families as well."
Sources: AAAS/Eurekalert! via University of Pennsylvania, eLife