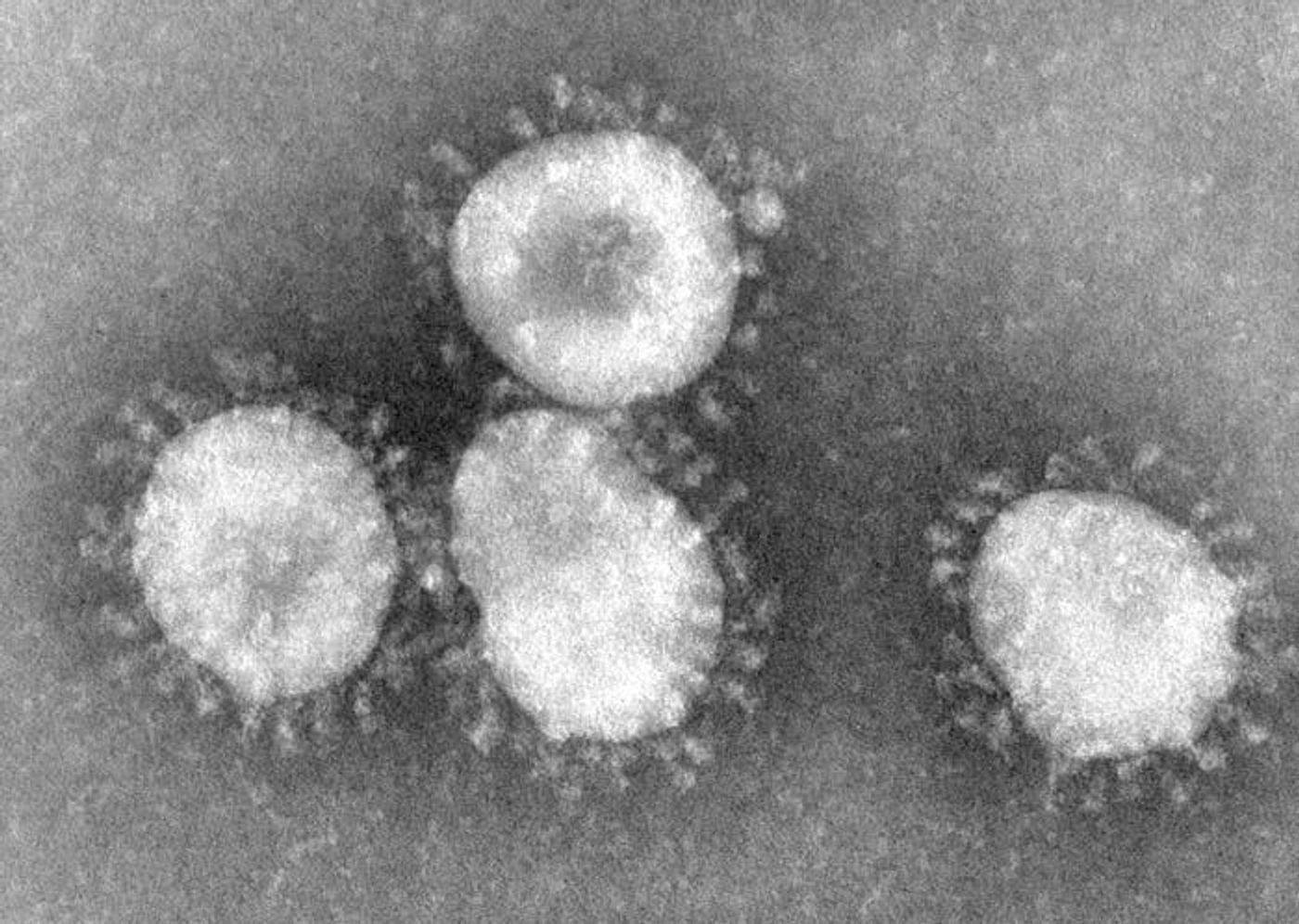
After a cold virus (rhinovirus) gets into our bodies it must get access to our cells in order to release its RNA and multiply. However, the mechanism by which the RNA is transferred is not well-understood or easy to study. Researchers at The Vienna University of Technology or TU Wien have created a new method to investigate this process that takes advantage of two established procedures, capillary electrophoresis on chips and molecular beacons.
Their study is published in the journal of Analytical and Bioanalytical Chemistry.
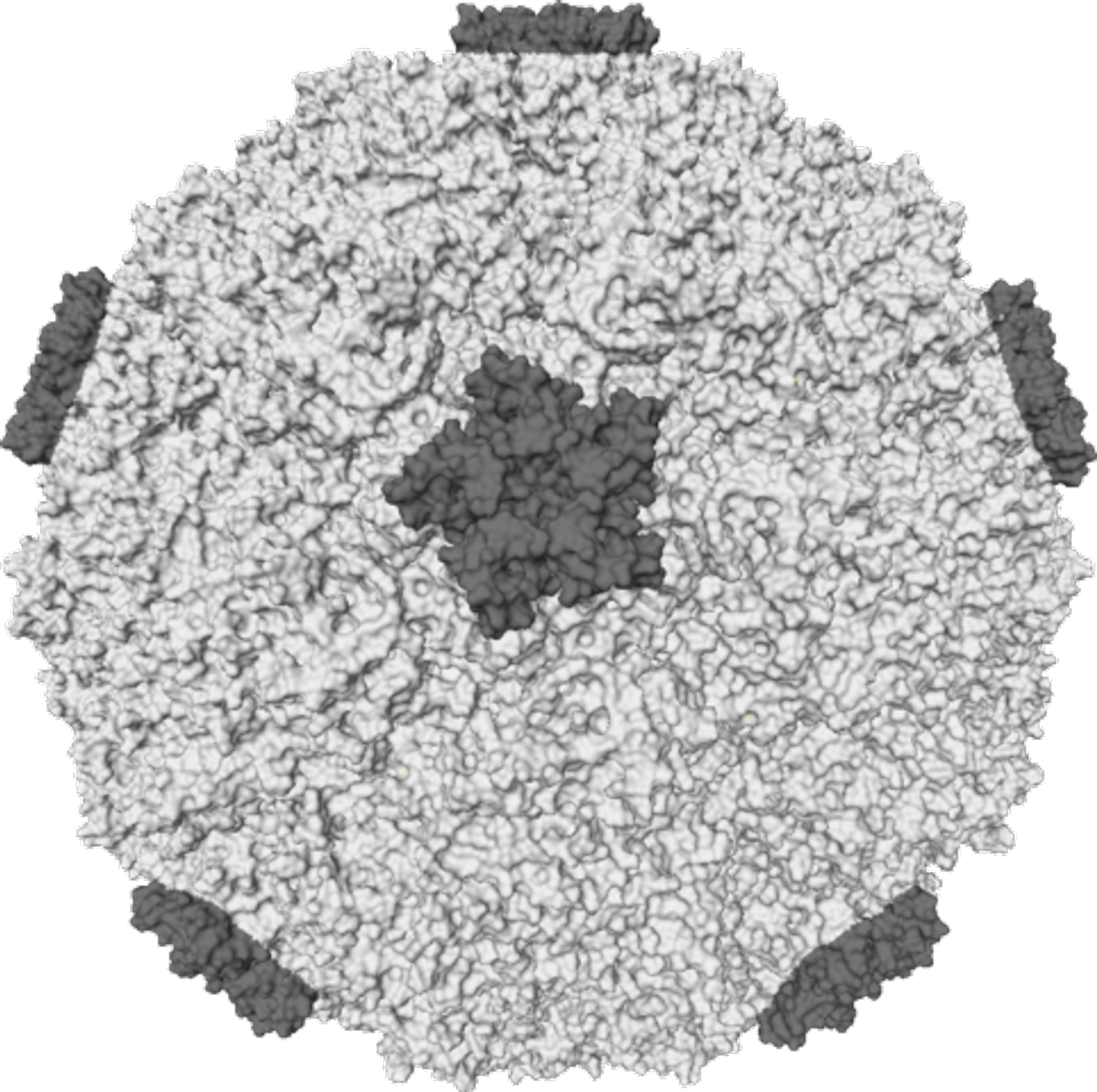
The structure of a rhinovirus is relatively simple, consisting of a shell or capsid made of four different proteins with 60 of each that are arranged in an icosahedron. The viral RNA is contained within it.
“Certain external conditions can also cause the virus to release its RNA to the outside,” says Victor Weiss, first author of this study. “In our cells this is triggered by a lower pH value; you can also achieve the same effect by increasing the temperature to 57 °C for ten minutes”. In this particular case, the proteins first rearrange themselves; the capsid of the virus then forms holes from which the RNA is released.
This type of in-depth understanding is critical for a number of medical reasons including for drug development that would prevent the RNA release. The new technique has made it possible to view the dynamics of this process directly.
Molecular beacons were used for this work. They are specialized molecules of RNA or DNA that have a fluorophore at one end and a quencher at the other. The fluorophore will flash if light of a particular wavelength - a laser - is shined on it while the quencher prevents the flashing. Victor Weiss explains, “To begin with, the molecule is folded up; the fluorophore and quencher are positioned very close to one another, then the fluorescence is very low.”
These beacons can be made to hybridize to a very specific sequence of RNA. When that happens, the molecule unfolds and the fluorophore and quencher are suddenly far apart from one another. When a suitable laser light is shined on the molecule, it lights up. Thus, molecular beacons can verify an RNA sequence.
The second proven technique that was used in the study is capillary electrophoresis, separation of components of a sample by moving it through an electric field. The final procedure works like this: a small sample of liquid is put in a channel where an electric field is applied. Various nanoparticles then migrate at different speeds based on their makeup. After a separation distance of about one and a half centimeters, a laser beam strikes the particle. The unfolded molecular beacon that is attached to the viral RNA lights up, and the fluorescence is measured.
"The different components of the sample reach the laser at different times. This is the only way to be sure that you are actually measuring exactly what you want to measure," explains Günter Allmaier, director of TU Wien and senior author of the study. "We can now demonstrate, for example, from which end of the RNA the virus first emerges, and how this process actually works."
“To us it's about developing the method; as a test object, the cold virus is virtually ideal," continues Allmaier. "However, we do of course hope that this method is established in medical research. We have now shown what great potential it has and this is also apparent in the partnership with Agilent Technologies."
Sources:
AAAS via
TU Wien News,
Analytical and Bioanalytical Chemistry