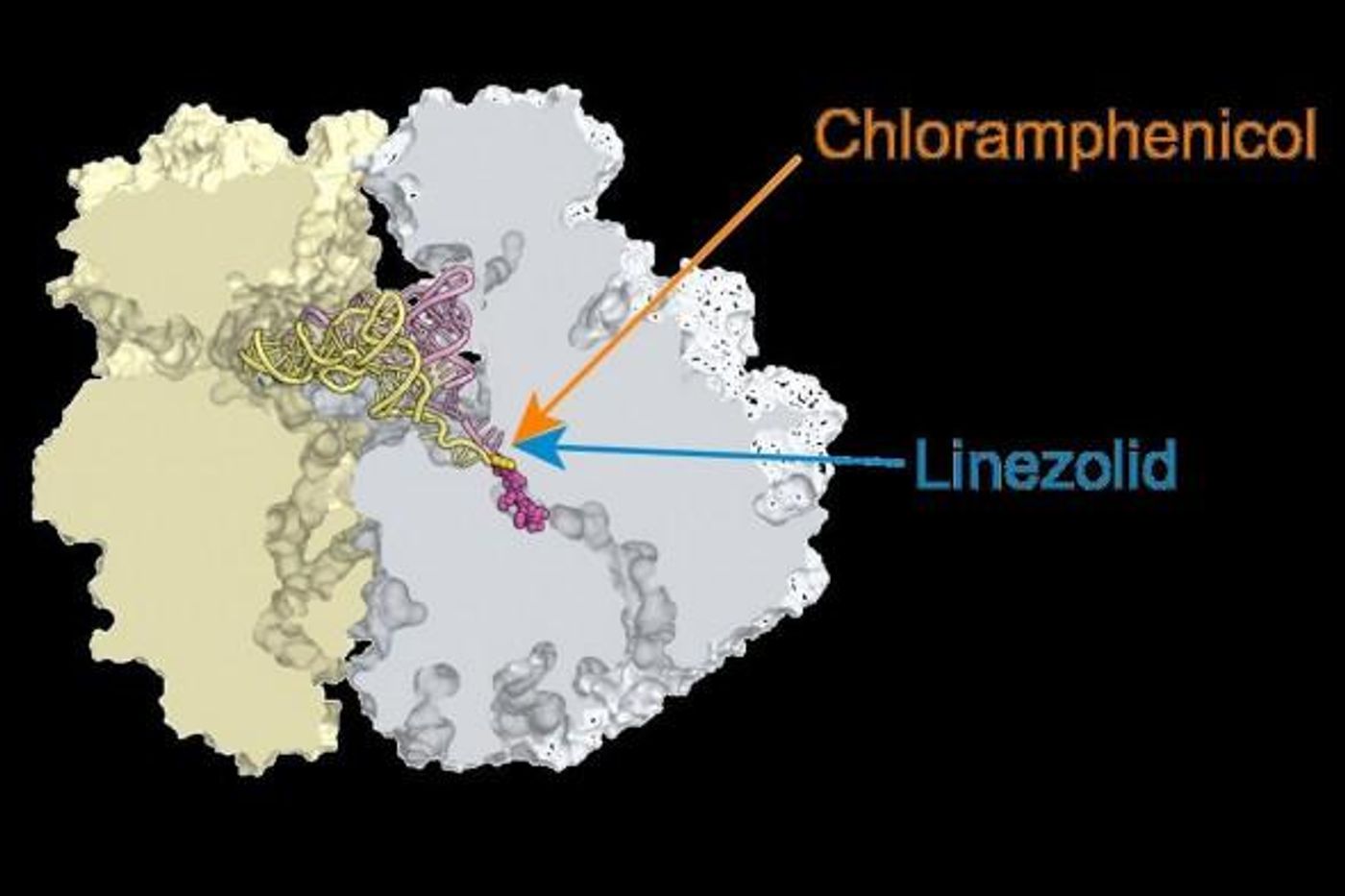
Researchers at the University of Illinois Chicago (UIC) have learned more about how two common antibiotics, chloramphenicol and linezolid, work. This new mechanism challenges what had been thought by doctors and scientists for many years as well. It seems that rather than the simply halting the creation of new proteins, the drugs stop the cell from synthesizing new proteins in very specific places in the genome.
The cell has a special organelle to carry out the manufacture of all the new proteins it needs, the ribosome; featured in the video below. It’s a complex piece of cellular machinery that is a typical target of many antibiotics. The research team behind the work, led by Alexander Mankin and Nora Vazquez-Laslop, has been conducting research on antibiotics and ribosomes. Using a combination of genomics and biochemistry they have learned more.
Their new publication in the Proceedings of the National Academy of Sciences reports that although chloramphenicol and linezolid target the ribosome at the catalytic center, the drugs put the brakes on protein synthesis only at specific sites.
"Many antibiotics interfere with the growth of pathogenic bacteria by inhibiting protein synthesis," explained Mankin, Director of the UIC Center for Biomolecular Sciences and Professor of Medicinal Chemistry and Pharmacognosy. "This is done by targeting the catalytic center of the bacterial ribosome, where proteins are being made. It is commonly assumed that these drugs are universal inhibitors of protein synthesis and should readily block the formation of every peptide bond."
"But -- we have shown that this is not necessarily the case," said Vazquez-Laslop, research associate professor of medicinal chemistry and pharmacognosy.
Pharmacognosy is the study of medicinal drugs that come from natural sources like plants. Chloramphenicol is one such drug and among the oldest antibiotics available. For decades it has been used to battle bacterial infections from meningitis to cholera and typhoid fever.
Linezolid is a newer, synthetic antibiotic and used to combat serious Gram-positive bacterial infections such as those from MRSA or other drug resistant pathogens. Mankin's team has previously worked to show the site of action of linezolid.
The antibiotics both work by binding the catalytic center of the ribosome, where it was thought they prevented formation of the peptide bonds that link the parts of a protein together. Mankin explained that it was expected that the antibiotics just interfere with the enzyme of the catalytic center from working.
"Contrary to this view, the activity of chloramphenicol and linezolid critically depends on the nature of specific amino acids of the nascent chain carried by the ribosome and by the identity of the next amino acid to be connected to a growing protein," Vazquez-Laslop explained. "These findings indicate that the nascent protein modulates the properties of the ribosomal catalytic center and affects binding of its ligands, including antibiotics."
"If you know how these inhibitors work, you can make better drugs and make them better tools for research. You can also use them more efficiently to treat human and animal diseases," concluded Mankin. A lecture by Mankin on the "Modeling of Peptide-mediated Ribosome Stalling with Antibiotics" is featured in the above video.
Sources:
AAAS/Eurekalert! via
UIC,
PNAS