New research published in the Journal of Biological Chemistry outlines how certain cells in the intestine, enterocytes, respond to an encounter with a high amount of fat, such as what would come from consuming foods rich in cholesterol and triglycerides – which are found in animal and vegetable fats.
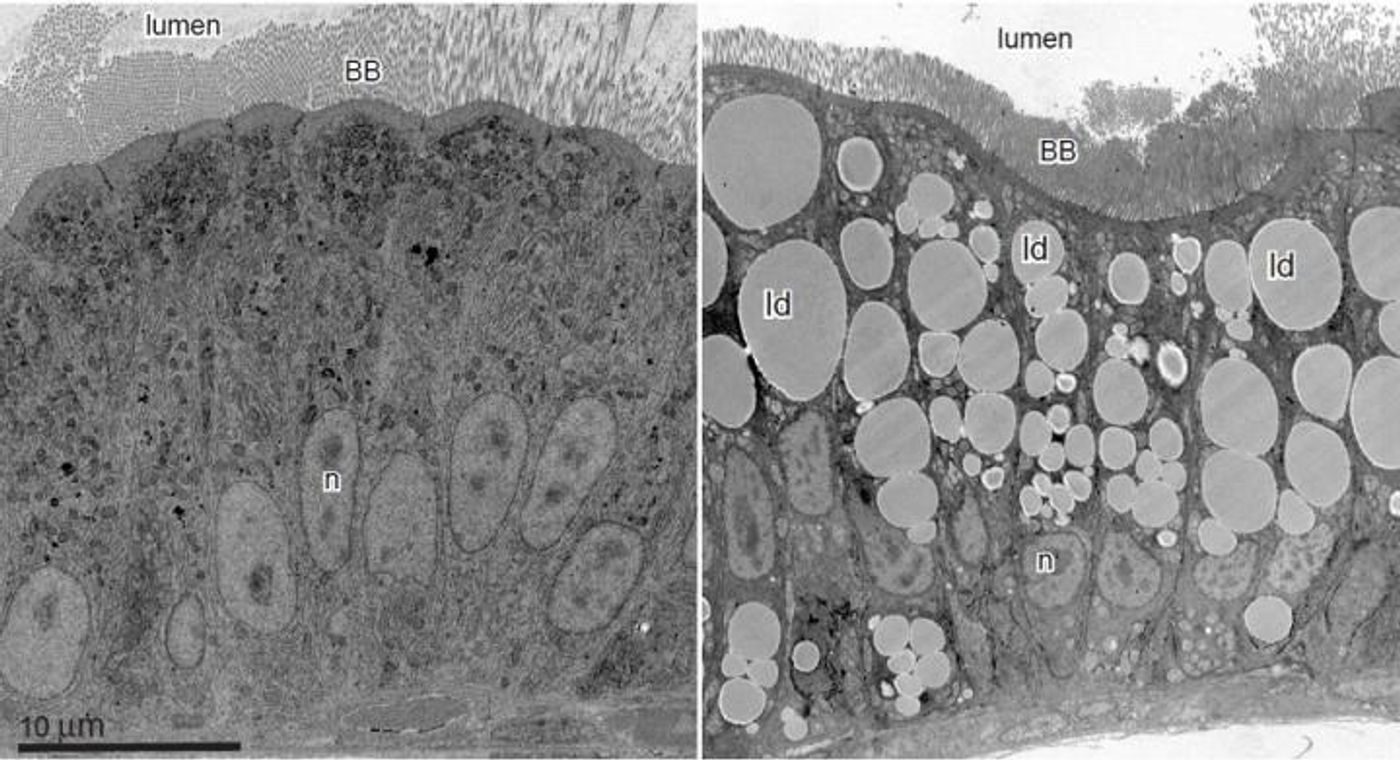
Inside of the intestine are enterocytes, and microvilli extend from the surface of the cells. The intestinal interior has many projections and cavities that increase the surface area of the intestine tremendously, allowing the body to maximize the absorption of nutrients. The enterocytes function to absorb and process the food during digestion. The video below has more on the process. One molecule that is taken up by the cells is a lipid from a fatty food. The researchers found that those fatty foods result in changes to the shape of organelles inside the enterocytes.
"When we eat fatty foods, our body's response is coordinated between our digestive organs, our nervous system, and the microbes living in our gut," said Steve Farber of the Carnegie Institution for Science in Baltimore. "Our research used zebrafish to focus on one aspect of this system--how the enterocyte cells inside our intestines respond to a high-fat meal."
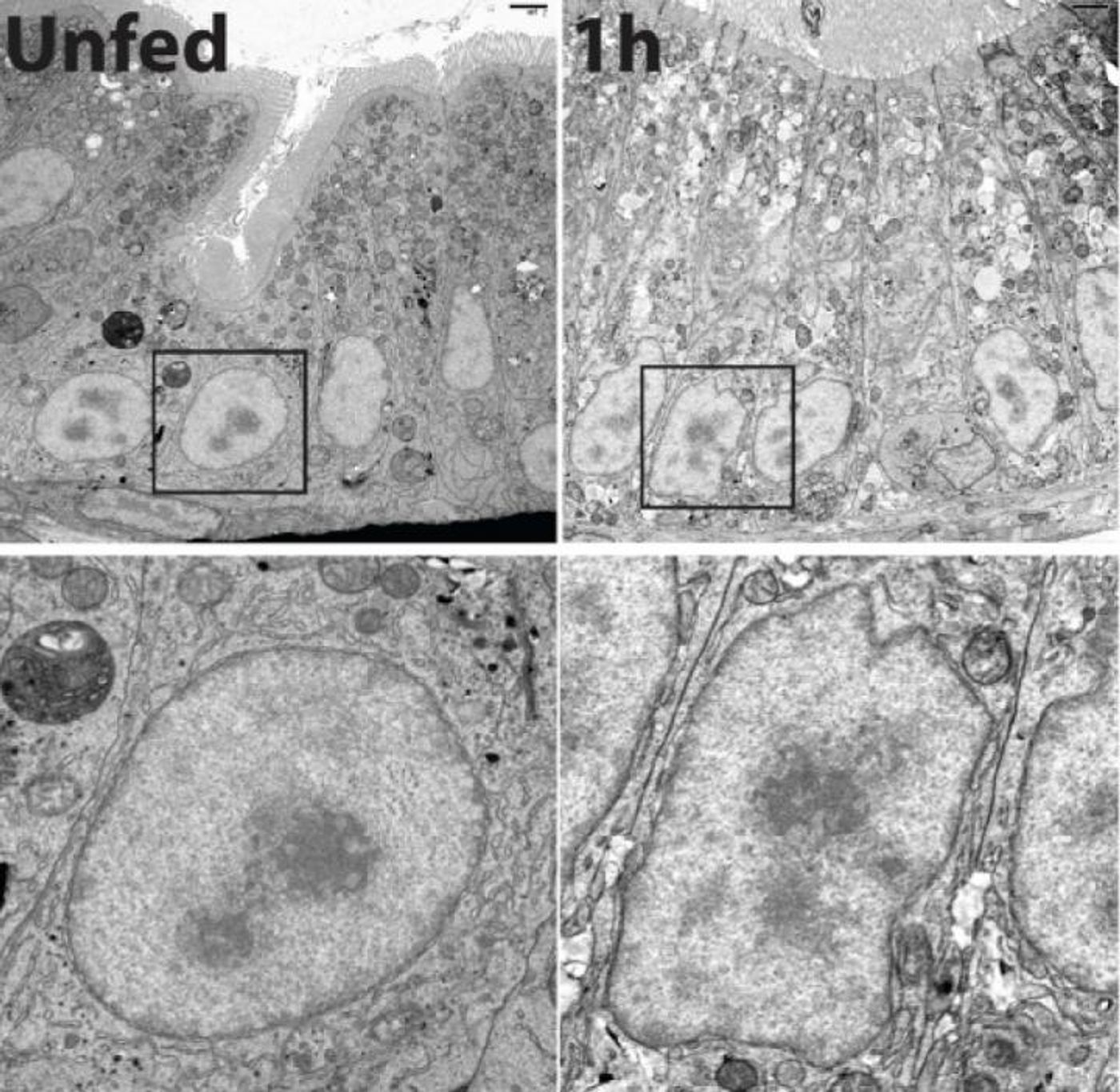
Farber’s team determined that the nucleus of a cell rapidly takes on a ruffled appearance following the consumption of fatty foods, a process that is reversible. That was of interest to the researchers especially because the nucleus houses the genetic material of a cell.
Delving deeper, the scientists learned that as the shape of the nucleus shifts, specific genes are also turned on, genes that regulate the packaging and distribution of lipids by the enterocytes. The researchers determined that that gene activation happens within only one hour of the high fat food consumption.
"Our working hypothesis is that the whole response to fat in the enterocyte--the remodeling and gene activation--may be coordinated by an organelle called the endoplasmic reticulum," explained the lead author of the work, Erin Zeituni.
The endoplsmic reticulum (ER) is another organelle of the cell, one that facilitates the assembly, packaging and distribution of the proteins needed by a cell. The researchers used drugs to interfere with a function of the ER. That function, the construction of lipoprotein particles, is needed to transport fats out of cells. When that interference occurred, the ruffling of the nucleus changed and for many genes, the activation process was disrupted. That suggested to the investigators that fat has an impact on the ER, which then initiates the intestinal response.
"So much of the process by which enterocytes prepare and package fats for distribution to the circulatory and lymphatic system is poorly understood. These findings should help increase our understanding of the basic molecular and cellular biology of intestinal cells," concluded Farber.
If you would like to learn more about how fat is processed by the body after a meal, check out the video above from the Khan Academy.
Sources:
AAAS/Eurekalert! via
Carnegie Institution,
Journal of Biological Chemistry