Complete BK Potassium Channel Structure Unveiled by Cryo-EM
Potassium channels are critical structures in biology; they selectively allow potassium ions to pass through the membrane surrounding cells. The precise gating of that channel is essential to its proper function; ion movement controlled by these channels is vital to electrical signaling in the body. New research from the team of Nobel Prize winner and Howard Hughes Medical Institute Investigator Roderick MacKinnon has finally revealed the complete structure of the BK ion channel. The work has been published in two Nature papers.
This work was made possible by new technology, cryo-electron microscopy. Old methodology required researchers to make crystals form from proteins, and could only elucidate their structure in that way. Large molecules like the BK channel, which investigators were unable to crystallize, often hinder that procedure.
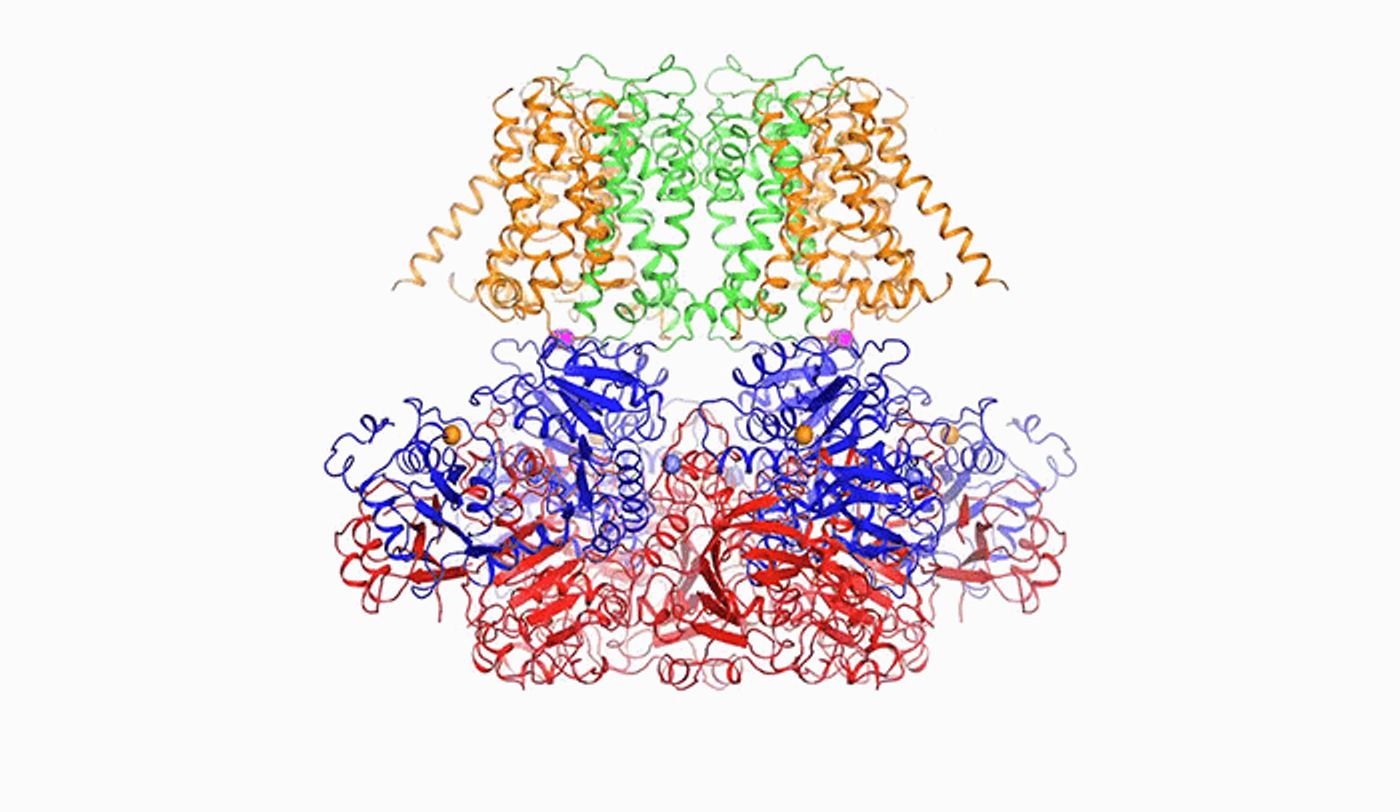
With cryo-electron microscopy, molecules can be frozen with ice instead of being crystallized. With this tool, the research team made a three-dimensional reconstruction of the atomic arrangement of the BK channel.
The new work provides a deeper understanding of the regulation of the channel by the cell; electrical signals are needed by the body in order for the brain to function properly and for moving muscles. The BK – big potassium – channel can move a lot of ions, and help to both regulate electrical signals and to stop them. The channel receives directions in two ways, from calcium levels, and electrical activity at the cell membrane. They were then able to look at the channel on an atomic scale, allowing them to see that the BK channel can transmit so many ions because of the large size of the channel passing through it.
“The pore within BK is much wider than those of other potassium channels. In fact, it forms a wide funnel that opens up to the interior of the cell,” said the lead author of one of the two new papers, Xiao Tao. “And the funnel’s surface bears a negative charge, which attracts positively charged potassium ions from within the cell toward the pore.”
It was found that when calcium ions (one type of signal) bind to the channel, the channels rearranges itself in a way that pulls some pieces outward, widening the opening of the pore. The researchers could not observe, however, how the channel acted when stimulated by a voltage change. The investigators suspect the channel can act is a specialized way to stimuli.
“Researchers have been looking to treat a number of conditions by developing drugs to activate BK, but without success so far,” said postdoc and lead author of the other paper, Richard Hite. “With the atomic structure of BK, we now have a map to guide these efforts by helping us understand which parts of the channel could be most effectively targeted.”
MacKinnon has been working on understanding the functions of this channel since his postdoc. “BK is the first potassium channel I studied, so it is enormously satisfying to finally observe its molecular structure,” concluded MacKinnon. Below, check out an interview with him from earlier this year.
A dysfunctional channel makes cells over active, as they are unable to stop electrical impulses. This can lead to a variety of diseases such as one form of asthma, hypertension, and a bladder problem in which the smooth muscles of the bladder are too active. A total understanding of its functions could lead to therapeutic avenues for treating dysfunction.
Sources: The Rockefeller University, Nature Hite et al 2016, Nature Tao et al 2016