High Level of Vitamin C Kills Cancer, may Improve Therapeutics
It was proposed as long ago as 1930 that vitamin C could be used to combat cancer, but that has remained a controversial idea. While there have been reports confirming that high doses of vitamin C is capable of selectively killing cancer cells in mouse models and cells in culture, researchers have not been able to determine the processes underlying this phenomenon. Scientists working at the University of Iowa (UI) have learned more about what vitamin C can do to cancer – if it is applied properly, as well as the likely mechanism. Previous work has examined the use of vitamin C as an oral therapy for cancer, and that could have been setting it up to fail.
A new report from the UI investigators shows that intravenous administration of vitamin C, which bypasses metabolic cascades in the gut that could send the vitamin to be excreted, elevates levels of the vitamin by 100 to 500 times what can be achieved by ingesting it orally. This super high dose is critical to the anti-cancer effect of the vitamin. The work is briefly described in the above video.
A redox biology expert at UI, Garry Buettner, has previously found that those highly elevated vitamin C levels will target and kill cancer cells while leaving healthy cells alone in mouse models and in the lab. Phase I traials have shown that the high level of vitamin C is safe in human patients, and improves outcomes. To see whether this could extend survival in people, doctors in UI hospitals and clinics are now proceeding to try the intravenous treatment in combination with typical radiation or chemotherapy.
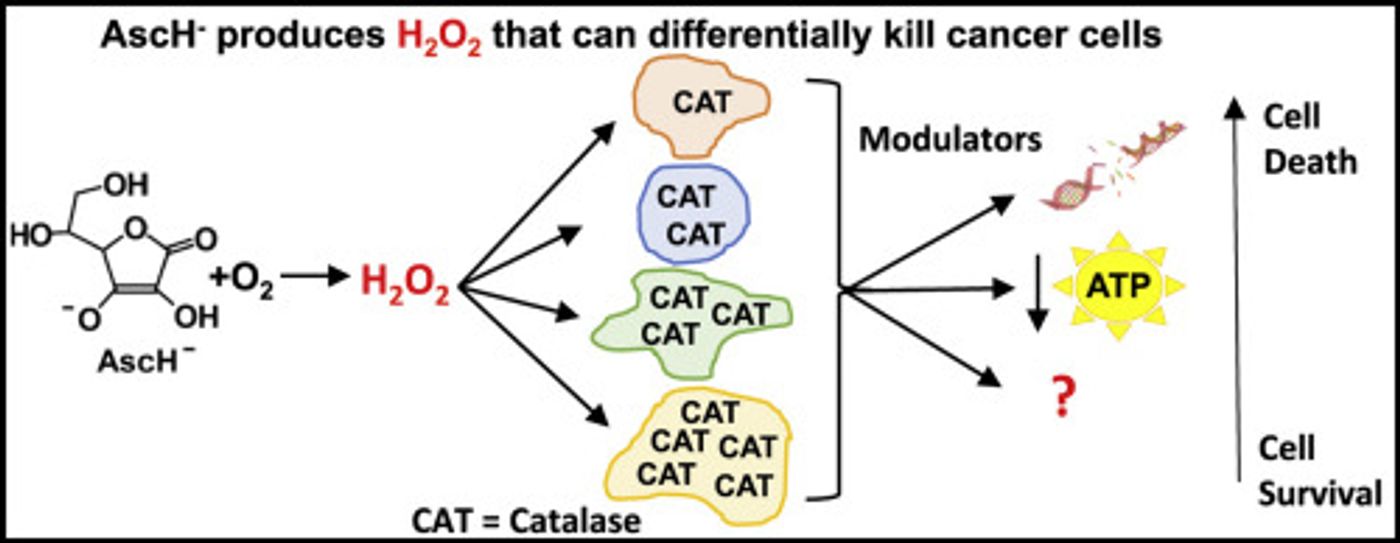
Reporting in Redox Biology, Buettner and colleagues have learned more about the mechanism underlying to effect. Vitamin C appears to easily break down, creating hydrogen peroxide in the process. Hydrogen peroxide is a reactive oxygen species, and they are known to damage cells and DNA. However, tumor cells are not very good at removing the hydrogen peroxide, unlike healthy cells.
"In this paper we demonstrate that cancer cells are much less efficient in removing hydrogen peroxide than normal cells. Thus, cancer cells are much more prone to damage and death from a high amount of hydrogen peroxide," explained Buettner, a Professor of Radiation Oncology and a member of Holden Comprehensive Cancer Center at the University of Iowa. "This explains how the very, very high levels of vitamin C used in our clinical trials do not affect normal tissue, but can be damaging to tumor tissue."
Normally, there are a few ways for cells to rid themselves of hydrogen peroxide, which will not cause damage if kept at a low level. This work indicates that an enzyme, catalase, is the tool used by the cell to remove hydrogen peroxide that is produced during the breakdown of vitamin C. It was found that in cells with less catalase activity, there is a greater likelihood of damage and death if they are exposed to high levels of vitamin C.
"Our results suggest that cancers with low levels of catalase are likely to be the most responsive to high-dose vitamin C therapy, whereas cancers with relatively high levels of catalase may be the least responsive," Buttner said. The researchers now hope to create a way to assay catalase levels in tumors. Bringing the therapy to tumors where it would be most effective could lead to better therapeutics for some types of cancer.
Sources: AAAS/Eurekalert! via UI, Cell Research, Redox Biology