Gut Microbes Drive Inflammation That Contributes to Premature Death
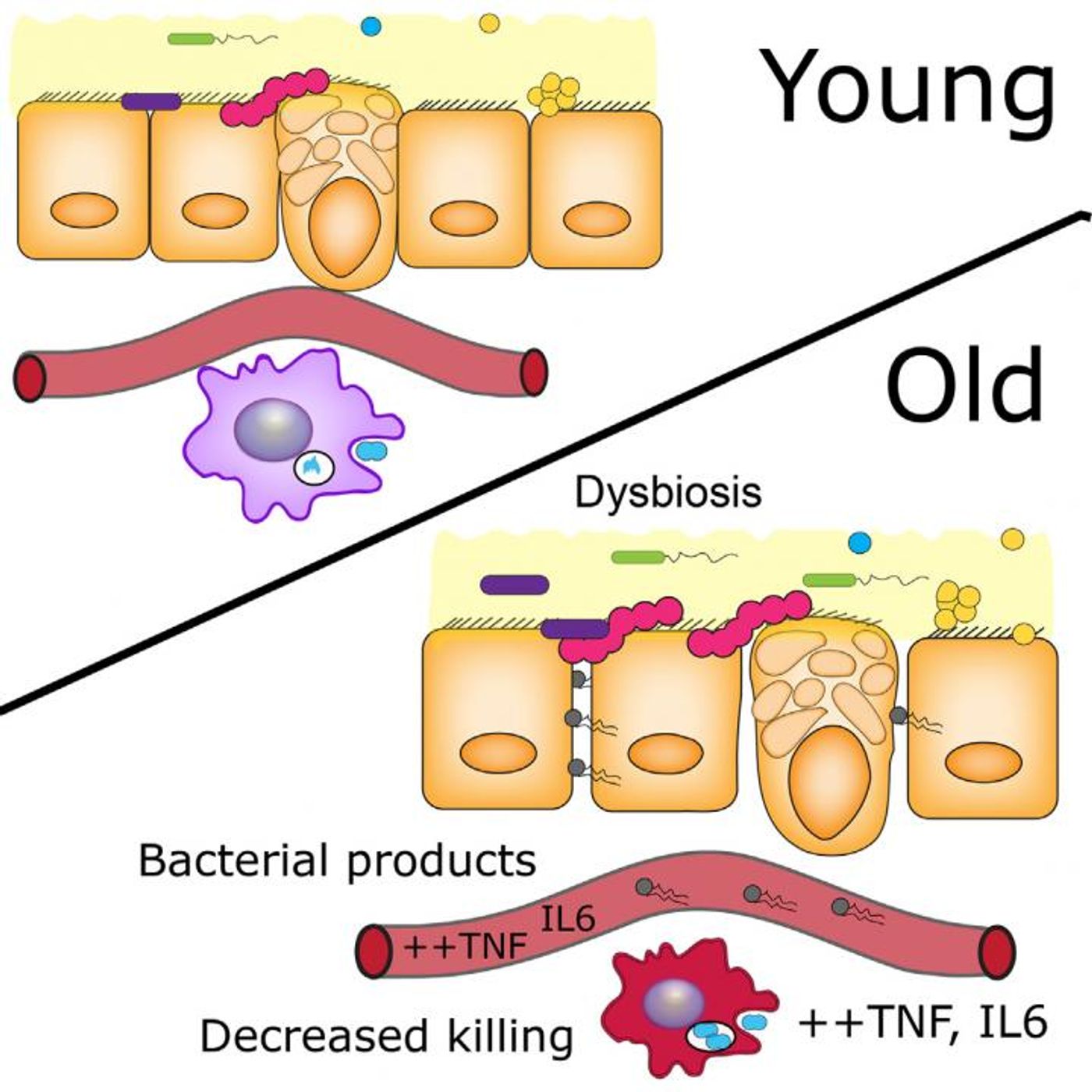
It’s known that the amount of inflammation-related molecules in the blood increases and becomes a strong risk factor for death as we age but it’s not known why. New research done with a mouse model indicated that gut microbes are one link between inflammation and premature death. As the gut microbes of mice became more imbalanced, the intestines of those mice got leaky. The weakness in the intestinal barrier allowed molecules made by the bacteria to escape from the gut and go on to trigger inflammation, impairing immune function. The work was reported in Cell Host & Microbe.
"To date, the only things you can do to reduce your age-associated inflammation are eat a healthy diet, exercise, and manage any chronic inflammatory conditions to the best of your ability," explained senior author Dawn Bowdish (@MsMacrophage) of the Michael G. DeGroote Institute for Infectious Disease Research at McMaster University. "We hope that in the future we will be able use drugs or pre- or probiotics to increase the barrier function of the gut to keep the microbes in their place and reduce age-associated inflammation and all the bad things that come with it."
Inflammatory biomarkers that are known to increase with increasing age include pro-inflammatory cytokines like tumor necrosis factor (TNF) ; when people exhibit high levels of these molecules, they’re more likely to be frail, hospitalized, or die. Such individuals are also less independent and more susceptible to certain types of infections; they have a range of diseases of old age like dementia and cardiovascular disease. While there have been suggestions as to how these conditions are linked, such as increased inflammation because of prolonged wear and tear on cells of the immune cells. Another hypothesis says it’s caused by immune cells that are engaged in battle with chronic viral infections. There has been little evidence to settle the debate, or give other insight.
For this work, the investigators used mice without microbes, germ-free mice. Bowdish and her colleagues raised them in an environment without germs, then compared them to mice raised normally. Interestingly, the germ-free mice did exhibit the typical symptoms of aging seen in inflammation like increases intestinal permeability, levels of bacterial products or pro-inflammatory cytokines in the bloodstream, compared to normal mice. The germ-free mice tended to live longer as well, to 600 days, while their aged macrophages maintained anti-microbial function. These findings indicated that changes in the gut microbiome due to age can indeed weaken the intestinal barrier, subsequent release of bacterial byproducts leads to a domino effect promoting inflammation, impairing immune function, and reducing lifespan.
Additionally, the scientists showed that inflammation and the microbiome exert effects on one another. TNF-deficient mice are protected from inflammation and no age-related changes in the makeup of gut microbes were seen. Treatment with an drug that interferes with TNF was able to reverse age-related changes in the microbiome.
"We assume that this is because if we reduce inflammation, we improve immune function, and if we improve immune function, we maintain the ability to farm a healthy gut microbiota, but we don't know for sure yet," Bowdish explained. "We also believe that targeting age-associated inflammation will improve immune health and are investigating repurposing drugs that are already on the market and developing novel strategies or therapeutics to this effect."
This work may help doctors use the microbiome and make the proper changes to it in order to improve the health of the intestine and decrease inflammation associated with age. "Since age-associated inflammation is linked to so many aspects of unhealthy aging, we predict that these strategies could help keep us healthy, active, and independent as we age," Bowdish said.
Check out the video for more information on how the microbiome influences health and disease. It features Claire Fraser, an expert in the field who was not involved with this work.
Sources: AAAS/Eurekalert! via Cell Press, Cell Host & Microbe