Female Mouse Embryos Actively Eliminate Male Tissue
It has long been taught that embryos will grow into a female unless male hormones called androgens alter the course of development and create a male. New work has challenged that idea, identifying a protein that can control whether a mouse embryo will develop a male reproductive system. The discovery has been reported in Science and is outlined in the following video.
Researchers at the National Institute of Environmental Health Sciences (NIEHS), part of the NIH, and at Baylor College of Medicine, led by Humphrey Hung-Chang Yao, Ph.D., studied the mechanism behind the development of female and male sex-specific reproductive systems. In the very early stages of growth, all mammalian embryos of both sexes have a reproductive tract structure for both genders. One must disintegrate, said Yao, for a mouse or a human to become female or male.
"I learned in graduate school that androgens are needed to maintain the male reproductive tract, but our work finds that maintenance of the male reproductive tract can be achieved without androgen," said Yao.
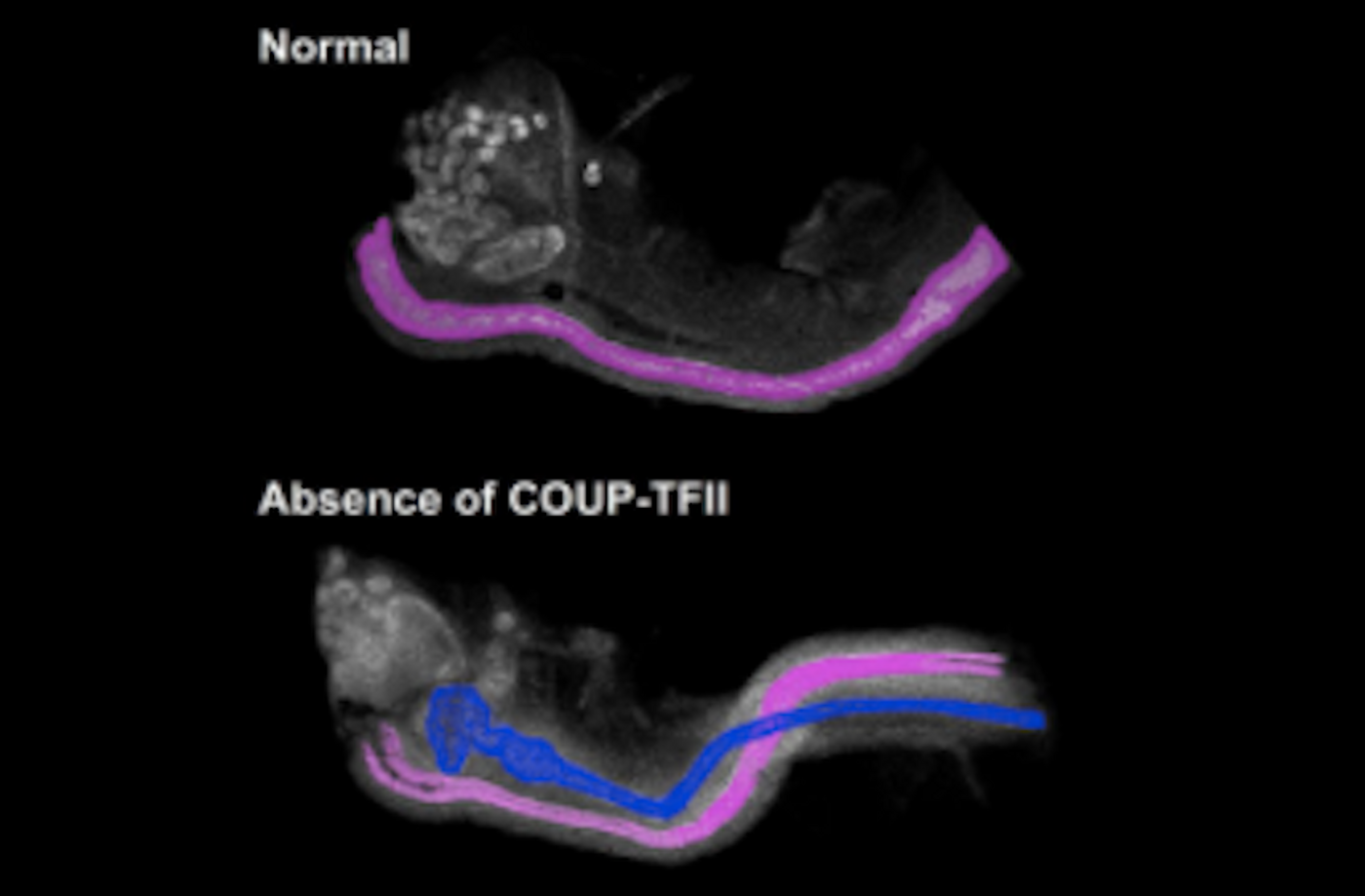
The protein responsible for choosing the gender fate of a mammal, the researchers found, is COUP-TFII. For this work, the team used a mouse model that ablated COUP-TFII in a developmental structure that matures into the male and female reproductive ducts. Using this kind of female mouse, Yao and lead author Fei Zhao, Ph.D. were quite surprised to see that it created both male and female reproductive ducts. “We were just scratching our heads,” said Yao, who is the head of the Reproductive Developmental Biology Group.
To explain these findings, the scientists checked to see if the ovaries had started to make testosterone. But no evidence of androgen synthesis was seen in the female mice that lacked COUP-TFII. The ovaries were normal. It was concluded that the male tract had developed without any androgen present.
It has been thought since the 1950s that embryonic testes made androgens that protected the male reproductive tract from being eliminated. Embryos became female simply by default, and the male reproductive tract broke down. This work suggests, however, that COUP-TFII is essential for the removal of the male tract, upending the old theory.
"This work is just the beginning, and many interesting questions remain unanswered," Zhao added. "We will continue to study how the embryo develops a functional reproductive system."
The group plans to continue this work to learn more about the origins of disorders of the reproductive system. They are collectively termed disorders of sexual development (DSD), including common defects like undescended testicles, or the genetic disorders Klinefelter Syndrome and Turner Syndrome.
"Individuals with DSD may have developmental challenges due to the presence of intersex organ systems," noted Kenneth Korach, Ph.D., head of the NIEHS Reproductive and Developmental Biology Laboratory. "With its highly novel approach and unexpected findings, Yao’s research has important implications for understanding the potential causes of these conditions."
Sources: NIH, Science News, Science