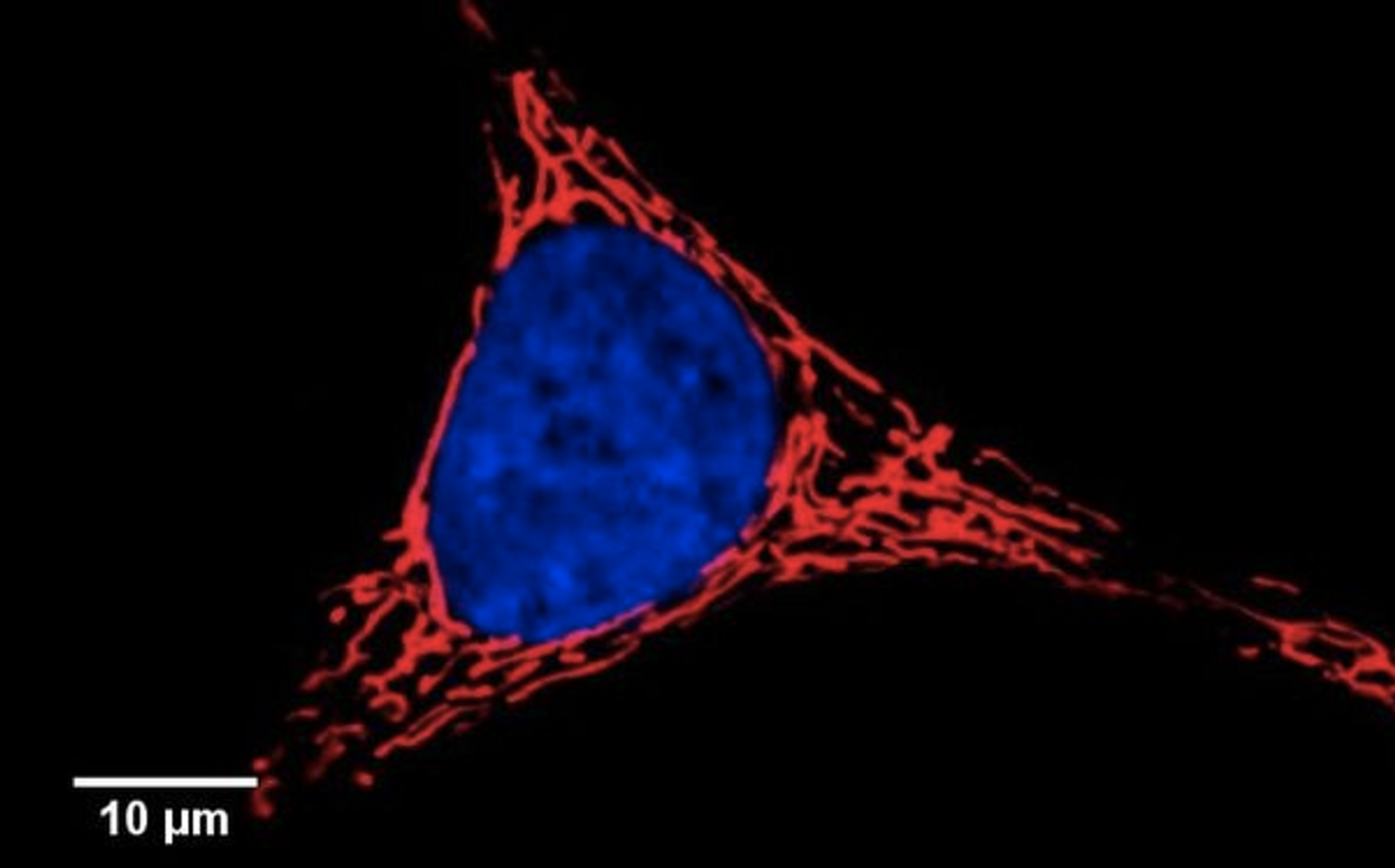
Cell Stress Elongates Mitochondria
Researchers have discovered a new pathway that promotes the activity of the cellular powerhouse, the mitochondria, during periods of stress; that could help protect against disease as we age. When our cells are impacted by stress, they halt protein production rather than create ones with mistakes. As that production stops, the mitochondria also change shape, becoming elongated. These findings, by scientists at The Scripps Research Institute, were published in the journal Cell Reports.
"Just a couple hours of not making proteins [are] to be enough to remodel the mitochondria, and they can stay that way for hours," explained the senior author of the research Luke Wiseman, Ph.D., associate professor at TSRI. "That seems to be a protective way to promote mitochondrial function during the early stages of stress."
The Wiseman lab studies how stress in another organelle, the endoplasmic reticulum (ER), impacts the shape and function of mitochondria. One part of this relationship is the Unfolded Protein Response (learn about UPR from the video), and a signaling pathway called PERK. A reduction in PERK signaling can cause dysfunction in mitochondria during stressful periods, disrupting cellular function. However, overactivity in the pathway can cause the cell to self-destruct. In an aging cell, keeping those things in balance becomes difficult.
"When you're older, little problems can become bigger problems because the PERK pathway isn't as good at responding," explained Wiseman.
Previous work has shown that PERK regulates many important aspects of mitochondrial function; it stops misshapen proteins from building up when the ER is under stress. This work indicates that stopping protein synthesis by activating PERK also affects the shape of mitochondria, increasing its length an changing its function.
The team wanted to know if cells were harmed or protected by these mechanisms, and how the energy output of mitochondria was impacted. Indeed, they saw changes.
"We were able to able to show a protective effect, where mitochondrial energy production was protected due to increased mitochondrial length" revealed co-first author Justine Lebeau, Ph.D. a research associate at TSRI.
This system may have evolved so cells could quickly respond to stress, the researchers suggested, when there isn’t time to generate proteins that can have protective effects.
"Blocking protein synthesis—and promoting cellular energy levels by regulating mitochondrial shape—seems to be an effective way of combatting stress over shorter time scales," said co-first author Aparajita Madhavan, a TSRI graduate student.
Wiseman suggests that when PERK sensitivity or activation are defective because of mutations or aging, mitochondria may lose protective regulation. Defects in PERK signaling have been implicated in illness, he added, like diabetes, heart disease, Alzheimer's and Parkinson's disease. He is hopeful that this research will help reveal therapeutic targets that might correct harmful mitochondria defects.
Sources: Phys.org via Scripps Research Institute, Cell Reports