The Neural Circuit Underlying Leptin has Been Revealed
Leptin is a molecule that is well known in obesity research; it is released by white fat cells, reducing appetite and contributing to the maintenance of healthy levels of blood sugar and weight. The mechanism behind these effects was not known, however. Now scientists have identified a neural circuit in the brain, specifically, the hypothalamus, that mediates leptin’s effects. This work, reported in Nature by scientists at the Tufts University School of Medicine challenges prevailing views about leptin. It may aid in the development of treatments for both types of diabetes and related complications.
When leptin or the receptors that it binds to are not properly regulated, extreme overeating from a raging appetite, obesity, and type 2 diabetes is the result. Leptin supplements are mostly useless, however; for reasons that are not understood, most overweight people are also resistant to leptin.
"While it's known that leptin receptors express in many neuronal types, extensive research has largely failed to uncover either a specific group of neurons that mediates leptin's primary effects or the molecular mechanisms involved. Whether such a specific group of neurons even exists is also controversial. Without identifying the real target that leptin works on, it is difficult to study its pathway or even effectively test any hypotheses," noted the senior author of the work Dong Kong, Ph.D., assistant professor of neuroscience at Tufts School of Medicine.
"Our mechanistic study provides important insight into the biggest issues - how leptin works and how leptin resistance develops - and making leptin a more clinically usable molecule to fight both obesity and diabetes. We also hope our research strategy and genetic tools will inspire researchers in other neurobiological and metabolic areas."
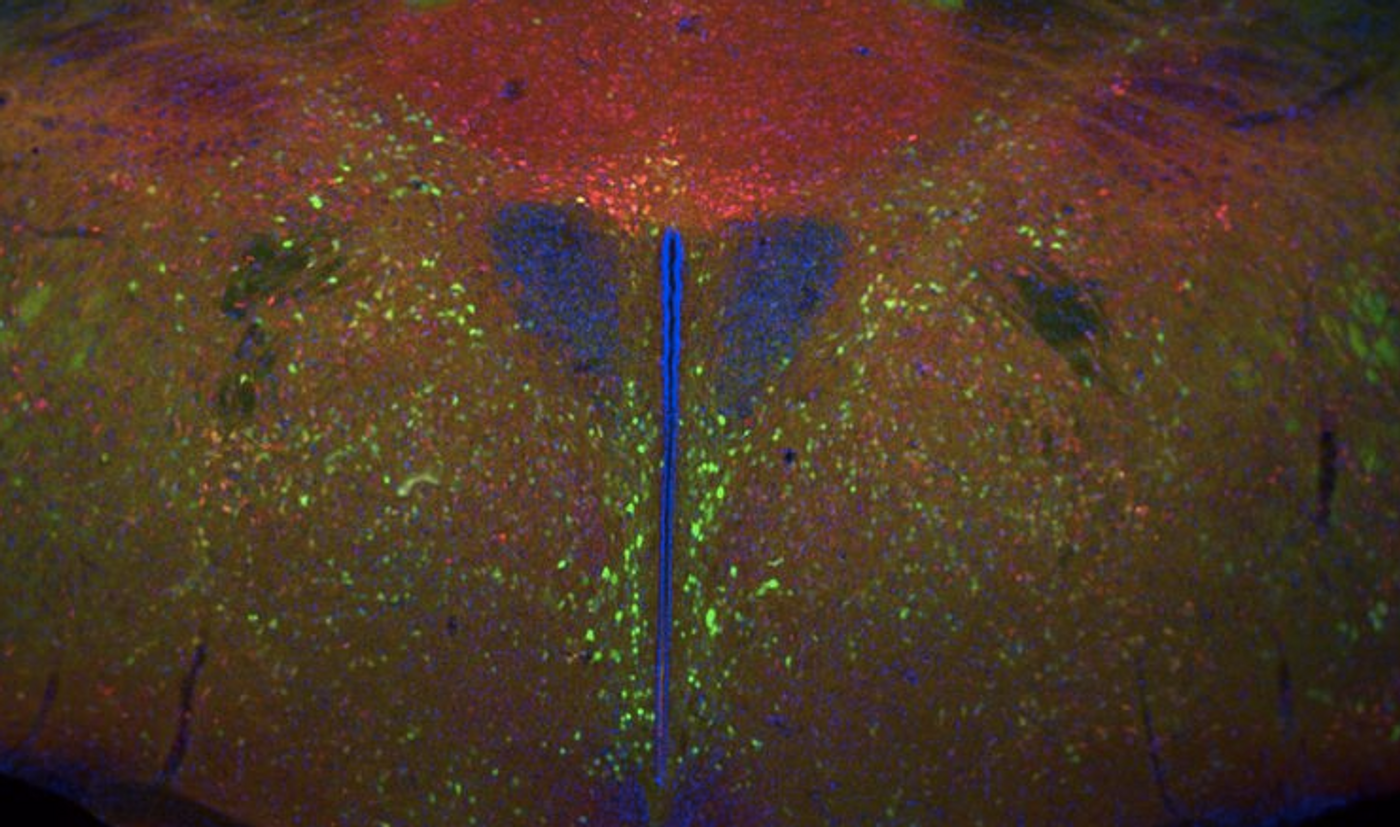
To find out how leptin works in the brain, Kong’s team started with the knowledge that leptin can rescue type 1 diabetes without the use of insulin, which bypasses leptin sensitivity. The team used streptozotocin (STZ), a drug to induce diabetes in mice that were not obese, as well as block the production of insulin and leptin. Then they mapped neuronal activity in the brain.
"We found that AgRP [agouti-related protein-producing] neurons in the hypothalamus were extremely active in these mice, and we suspected that the STZ-induced leptin deficiency was causing this. We were excited when we successfully used leptin to inhibit the AgRP neurons and quickly reverse the diabetes," noted the paper's co-first author Christopher Bartolome, a neuroscience graduate student.
Once proposed as a leptin target, AgRP neurons have since been written off by most leptin researchers because after leptin receptors were deleted specifically in AgRP neurons, the altered mice were not obese or diabetic like mice that have no leptin receptors. Kong’s team hypothesized that mice without leptin receptors are obese and diabetic at birth, which may have been masking the effect of leptin.
Using the CRISPR gene editing system, the team deleted leptin receptors from AgRP neurons in young adult mice. Targeting adult mice made this research different from previous work in this area.
"We found that deletion of leptin receptors in AgRP neurons induced marked obesity and diabetes and largely attenuated leptin's anti-obesity and anti-diabetes effects. This demonstrated that AgRP neurons represent the major site in the brain to mediate leptin's effects," said Kong.
Thus AgRP was pinpointed as a prime target of leptin in the brain. They also showed that two mechanisms are used by leptin to inhibit those neurons.
"These findings are very important because fully understanding the neural basis of leptin's effects could help lead to a better understanding of the causes of obesity, leptin resistance, and, even more importantly, to the development of mechanism-based therapies," said the co-first author of the report Jie Xu, Ph.D., a postdoctoral fellow in the Kong laboratory.
Learn more; check out the video above, an NIHvcast about leptin and the hypothalamic control of hunger.
Sources: AAAS/Eurekalert! Via Tufts University School of Medicine, Nature