Hypothalamic Neurons in the Obese are Different
The hypothalamus is a part of the brain that plays essential roles in hunger and satiety. Researchers have learned more about the neurons in that region. Scientists took skin and blood cells from healthy and very obese people and used them to create neurons that mimic those in the hypothalamus. The researchers found that when neurons are derived from cells of the obese, those neurons would not respond correctly to signals that indicate hunger or food intake; they showed differences in genes and metabolic pathways related to obesity. The data has been reported in Cell Stem Cell.
"This is the first step in applying an iPSC-based platform to model complex polygenic diseases such as obesity," said senior author Dhruv Sareen, a stem cell biologist at Cedars-Sinai Medical Center. "We developed a great platform that could potentially be used to evaluate the effects of experimental therapeutics on patient-specific hypothalamic neurons from obese patients with different, genetic backgrounds, body mass index, and environmental exposures."
Most people that are obese carry gene variations that make slight contributions to their susceptibility to weight gain; those genes are often related to metabolism and food consumption. The researchers wanted to learn more about how the hypothalamus contributes to obesity, but that (brain) tissue is difficult to obtain from people.
Sareen and colleagues overcame that hurdle by finding a way to make neurons like those in the hypothalamus from other cell types.
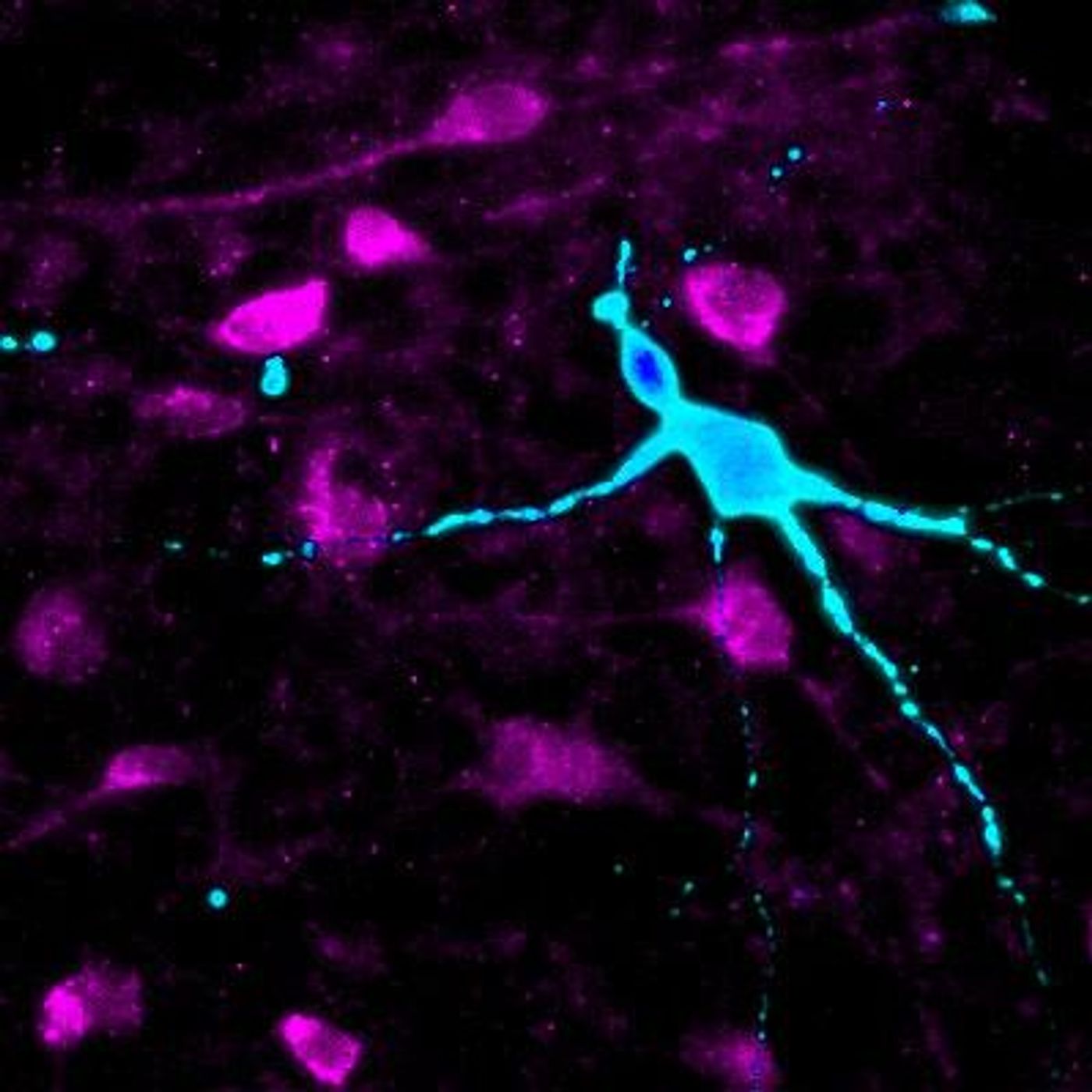
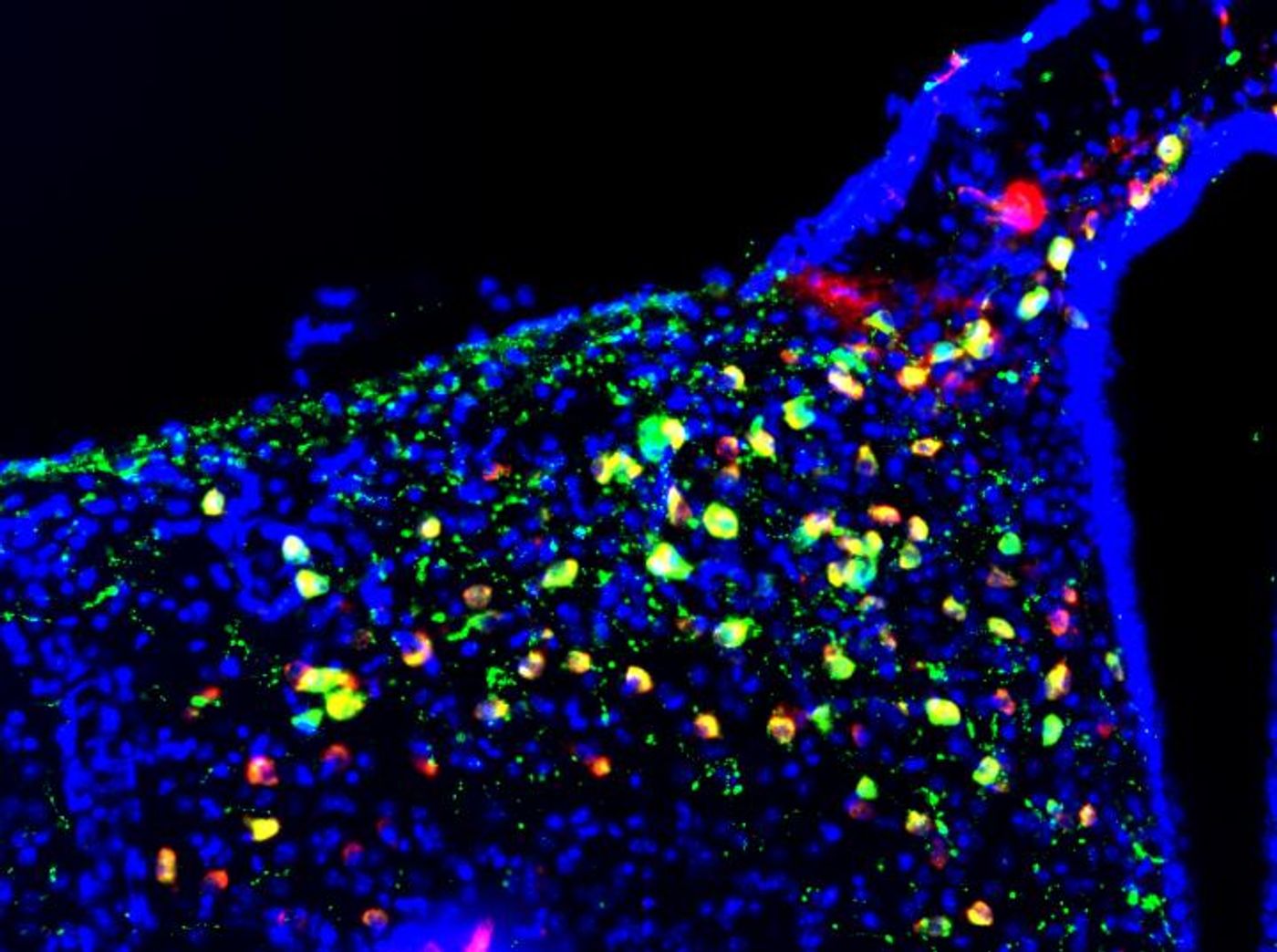
Their technique used blood and skin cells to generate hiPSCs (human induced pluripotent stem cells, described the video) from people with a body mass index (BMI) over 50, and those with a BMI under 25, considered normal weight. The hiPSCs then were utilized to make neurons with gene expression profiles that were similar to that of hypothalamus cells post-mortem (the profiles we have easy access to).
Those neurons release neuropeptides that respond to two hormones, ghrelin and leptin. In the neurons made from obese people, there was a stronger than typical reaction to ghrelin. The neurons also showed signs of disrupted metabolic pathways. These specialized hiPSCs are another step forward, showing how they can model disease in unusual or hard-to-obtain tissue types.
"Ultimately, we are paving the way for personalized or precision medicine, in which drugs could be customized for obese patients with different genetic backgrounds and metabolic disease status, depending on their tolerance and reaction to drug combinations," noted Sareen.
The hypothalamus does not regulate hunger on its own, however. Sareen and his team are aware of the limitations of their study, and in the future want to see how their hypothalamic-like neurons interact with other important cell types.
"There are several intermediate steps before we can realize the potential of this iPSC technology to become a reliable platform to screen potential therapeutics for obesity and other metabolic diseases,” added Sareen.
Sources: AAAS/Eurekalert! Via Cell Press, Cell Stem Cell