Ultra-fast Chemical Reaction that Turns Graphene into Semiconductors
German and US scientists recently reported an unusual feat: they observed the world's fastest chemical reaction, during which hydrogen atoms bind onto and then leave a sheet of graphene, all within ten quadrillionths (10^-14) of a second.
Since its discovery in the early 2000s, graphene has been under the limelight of the scientific community for being the material of the future. Although sharing a similar structure with other carbon-based material such as graphite and charcoal, graphene has a lot of unusual properties, steel-beating strength, and remarkable heat and electricity conductivity.
However, graphene isn't perfect: it lacks band gaps, a property that would give graphene the ultimate versatility in electronic and circuitry applications. A band gap is the energy needed for a valence electron to jump from one atom to another within the crystal lattice. The bigger the band gap, the harder for electrons to migrate and the less conductive the material is.
Materials that have large band gaps make good insulators, whereas those with very small or no band gaps are favorable conductors (such as pure graphene). Adding a small or big band gap to graphene can transform it into a semiconductor or an insulator, significantly broadening its usefulness in electronics and circuit boards.
A collaboration group between Max Planck Institute (MPI) and the California Institute of Technology (CalTech) intends to use hydrogen atoms to create a band gap in graphene. To achieve this, they relied on "bunch-compression photolysis", a technique first developed back in 2014 to conduct higher time-resolution experiments that involve reactions by atomic collision.
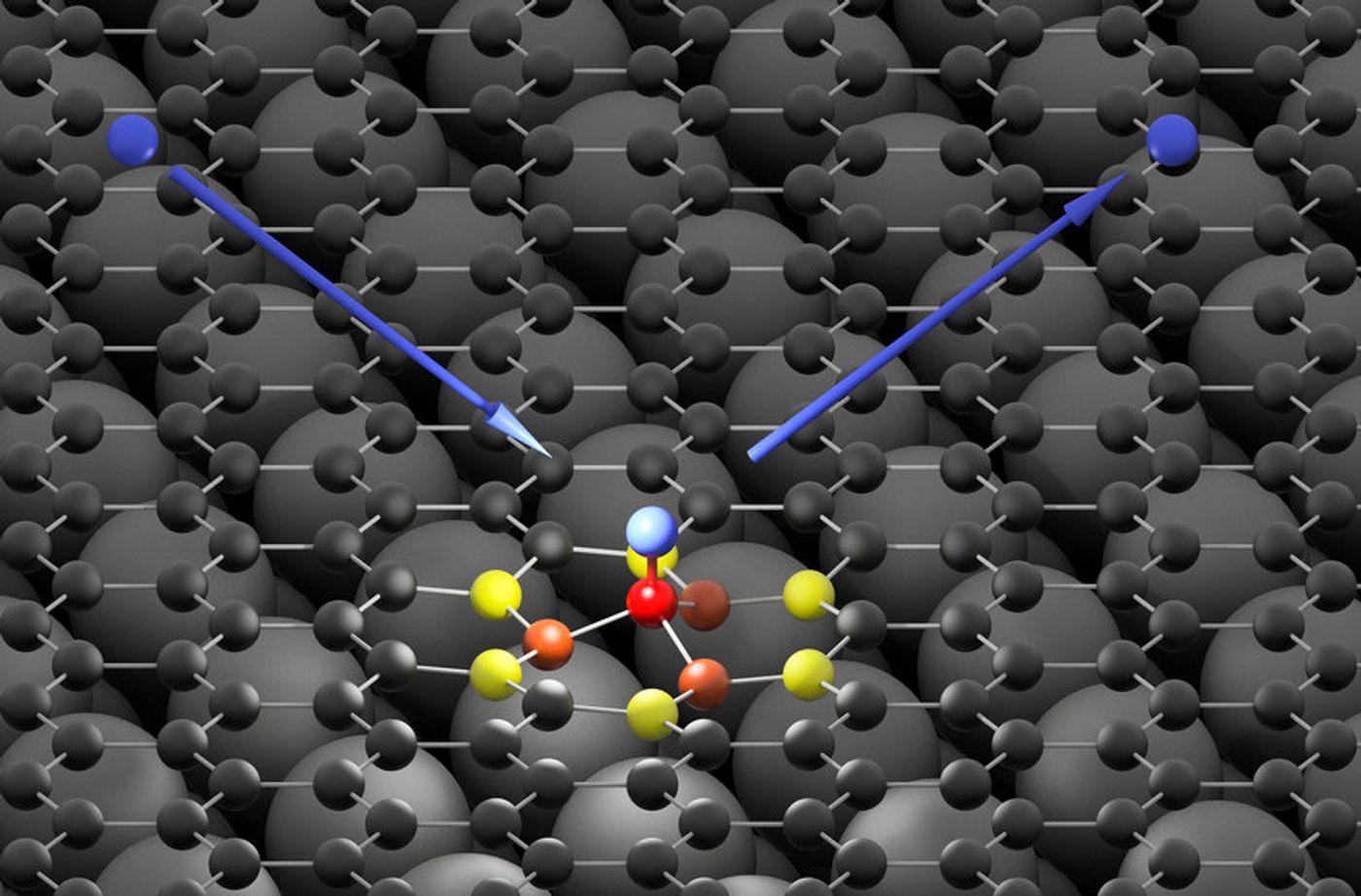
By bombarding a sheet of graphene inside a high vacuum environment with ultra-short pulses of hydrogen atoms, the researchers observed the scattering of hydrogen atoms from graphene. With the help of their highly time-resolved setup, they discovered that a transient carbon-hydrogen bond formation as the hydrogen atom lands on the sheet.
It was believed that the hydrogen atom transfers almost all of its kinetic energy to graphene upon collision, a phenomenon the researchers compared to a pebble hitting the water, which triggers a series of waves. Since the bond exists for an extremely short window, the hydrogen atom dissociates right away, and the bond dissipates.
Their results provide the chemistry community eye-opening insights into such type of chemical bonding. Scientists were quite surprised to find that the hydrogen atom can bind more quickly to the carbon atom, which contradicts predictions from the previous modeling.
This discovery was published in the journal Science.
Why graphene hasn’t taken over the world...yet (Verge Science)
Source: ScienceDaily