New Helium Compound Turns Chemistry Upside Down
We often wonder what life and environmental conditions would be like on other planets. Scientists have speculated scenarios both similar to what we have on Earth and also completely different from everything we know. The latter is hard to imagine, given we live within certain physical laws. If those laws are broken on other planets, what would happen?
Scientists have recently been studying high-pressure environments, such as those imagined to occur within the core of the Earth and on gas giants in our solar system. Last month, researchers from Harvard created never before seen metallic hydrogen using extremely high-pressure physics. This week, using a similar set-up, an international team of researchers worked together to create a compound with helium.
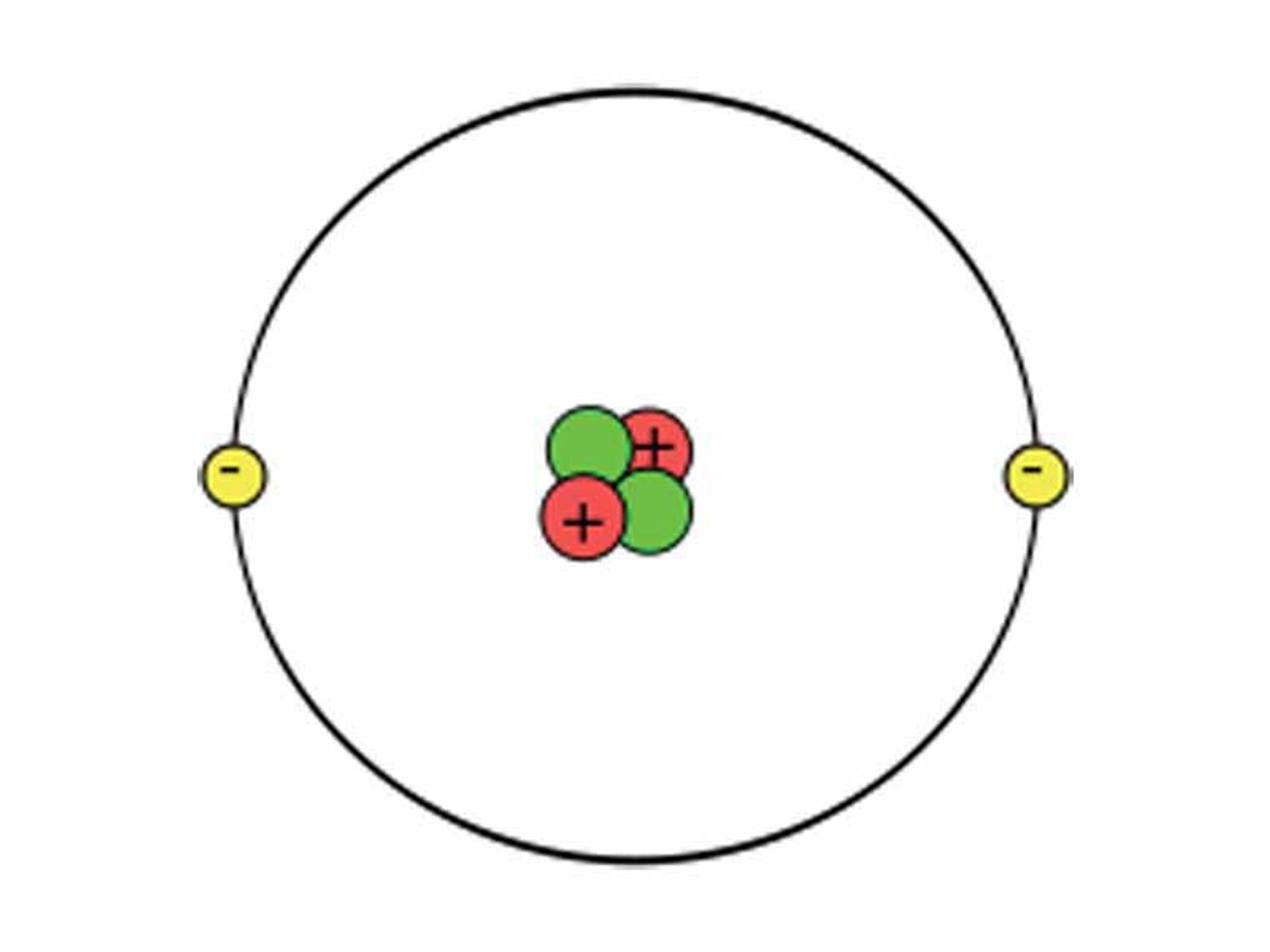
If you remember anything from chemistry in school, you would laugh and say it isn’t possible! Helium is a noble gas and does not create compounds with other elements. And under normal conditions you would be right. Helium is the first noble gas in the periodic table, made up of two protons and two electrons. With only a single shell holding its two electrons, the element is an inert, colorless, odorless, and very stable gas. It is the second most abundant element in the known universe, making up an estimated 24% of elemental mass on Earth, the sun and Jupiter.
Scientists have hypothesized for decades that chemistry in extreme conditions can challenge what we know of standard elemental laws. However, how can we test this when we live in a stable environment with consistent physical laws and conditions? To create an extremely high-pressure environment, like that thought to occur at the center of the Earth, researchers created a diamond anvil cell. This device can push atoms together so tightly between two treated diamonds that their properties can change.
In this case, the team was able to mix helium with sodium crystals to create a stable compound at extreme pressures. It is thought that helium compounds such as this one could possibly develop naturally on other planets with high pressure atmospheres, such as Saturn and Jupiter.
“Chemistry changes when you apply high pressure, and this can be achieved inside our Earth and on different planets like Saturn,” study co-author Ivan Popov, a doctoral student at the Utah State University, told Gizmodo. “But this is a book changer.”
The scientists note that the compound created does not contain real bonds as we know of in standard chemistry. Instead the compound is an ‘electride,’ where electrons are dispersed within a crystal structure. The experiment provides insight into what other structures and compounds might exist in high-pressure environments that we don’t observe here on Earth, and also could possibly revise chemistry as we know it.
Sources: Gizmodo, Nature Chemistry