This Solar Energy-powered Sponge Can Turn Carbon Dioxide into Fuel
Schematic showing how a spongy photocatalyst converts carbon dioxide into carbon monoxide. Credit: K. Niu & H. Zheng (Berkeley Lab)
In light of the warming climate, many countries around the globe are doing their best to curb carbon dioxide emissions. According to the Paris agreement, member states promised to reduce their carbon output to keep global warming to well below 2 degrees Celsius or 3.6 degrees Fahrenheit by the end of 21st century.
While limiting emissions is one way to achieve the goal (which appears to be less and less reachable), removing carbon from the Earth's atmosphere is considered a critical supplementary method. Many so-called negative emissions technologies are under development, and many hope that they can become widely deployed soon. Besides the already available carbon capture and storage technique, other strategies such as enhanced weathering within agriculture, carbon capture with ocean thermal energy, and bio-energy with carbon capture and storage have been gaining traction.
Related: Pulling carbon from the air
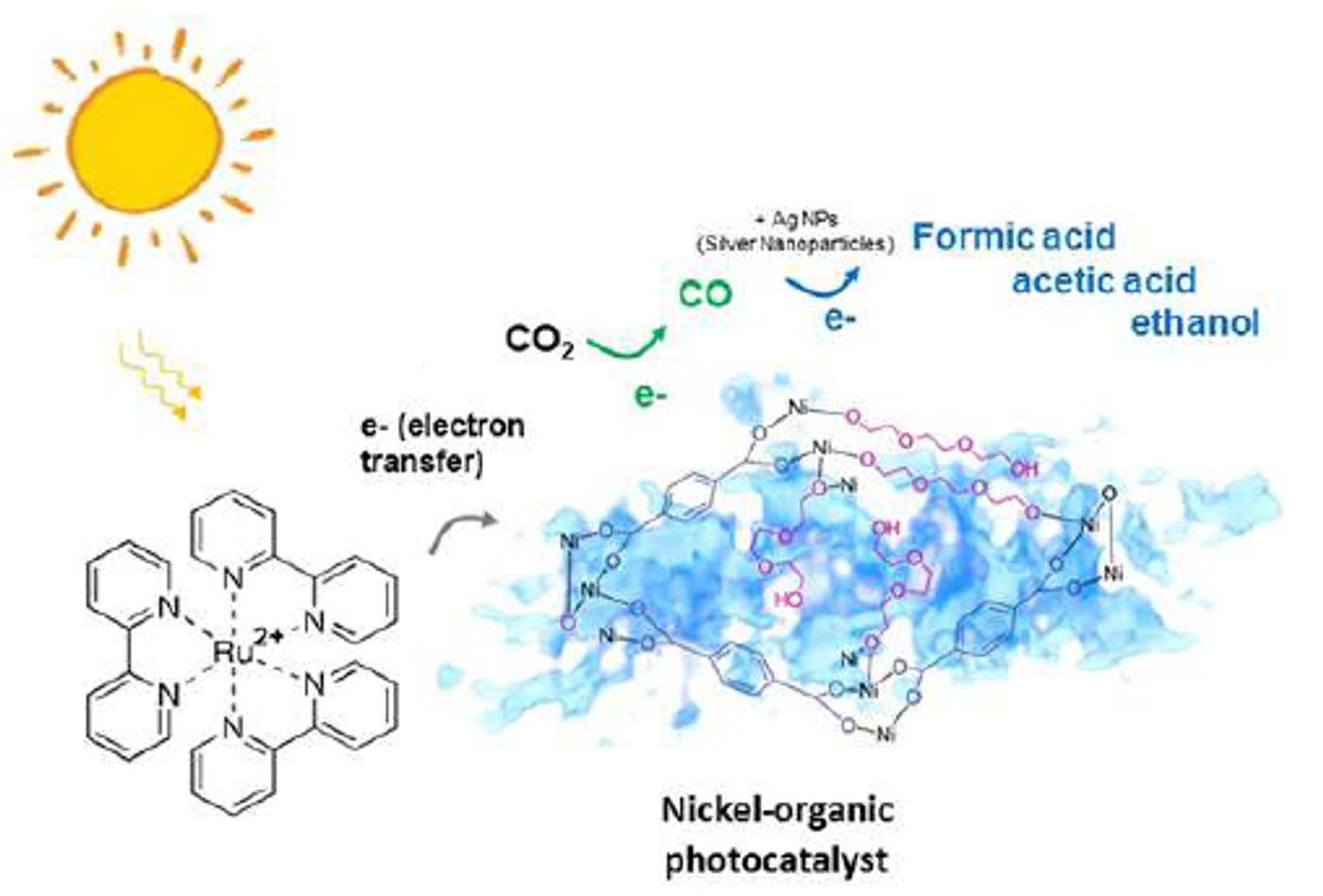
According to a recent publication in Science Advances, a joint research team from the US and Singapore has come up with a photocatalytic material that can turn carbon dioxide into carbon monoxide using solar energy. This nickel-organic composite can achieve what plants do with photosynthesis in a sense. Chemically, photosynthesis is a reduction reaction, a reaction in which the product gains electrons. Plants (as well as most algae and cyanobacteria) strip electrons from water and pass them onto carbon dioxide, producing carbohydrates (as their source of energy) and oxygen. This photo-activated material can turn CO2 into CO, which can further be transformed into fuel molecules with ease.
Eliminating competing chemical reactions in the reduction of carbon dioxide is the main battle to convert CO2 to CO because the involvement of hydrogen leads to by-products. The key of the team's success was a metal-organic composite made of nickel and triethylene glycol through exposure to an infrared laser. By changing the wavelength of the laser, the researchers obtained different composites, among which light-activated candidates were selected and heat-activated ones were screened out. While the nickel-organic photocatalyst resembles an average metal-organic framework, it also has a mix of soft linkers of varying lengths connected with nickel, creating a more porous space (like a sponge) to allow catalysis to happen. The photocatalyst showed high efficiency in converting CO2 to CO, with a production rate of ~1.6 × 10 μmol per hour per gram of CO2. More importantly, the conversion did not produce any hydrogen gas as by-product. As a second part of the test they enriched the catalytic composite with rhodium or silver nanocrystals, which drives the reaction toward producing formic acid and acetic acid.
The group recognizes that this technology can be used to generate fuel and other energy-rich products while mitigating the level of carbon dioxide. “The world right now is in need of innovative ways to create alternatives to fossil fuels, and to stem the levels of excessive CO2 in the atmosphere. Converting CO2 to fuels using solar energy is a global research endeavor. The spongy nickel-organic photocatalyst we demonstrated here is a critical step toward practical production of high-value multi-carbon fuels using solar energy," said Dr. Haimei Zheng the senior author and a principal investigator in the Lawrence Berkeley National Laboratory.
Carbon capture and storage. Credit: New York Times
Source: Science Advances/phys.org