The Intrinsically Fluorescent Nature of GFP Chromophore
Proteins from the green fluorescent protein (GFP) family are essential markers in the world of molecular and cellular biology because they help to visualize cellular components and processes. They fluoresce thanks to a small light-adsorbing fragment of the protein (a chromophore group). For a long time, scientists have had the perception that the chromophore emits light only in its natural protein environment. Once the fluorescent protein denatures, its chromophore loses its fluorescence.
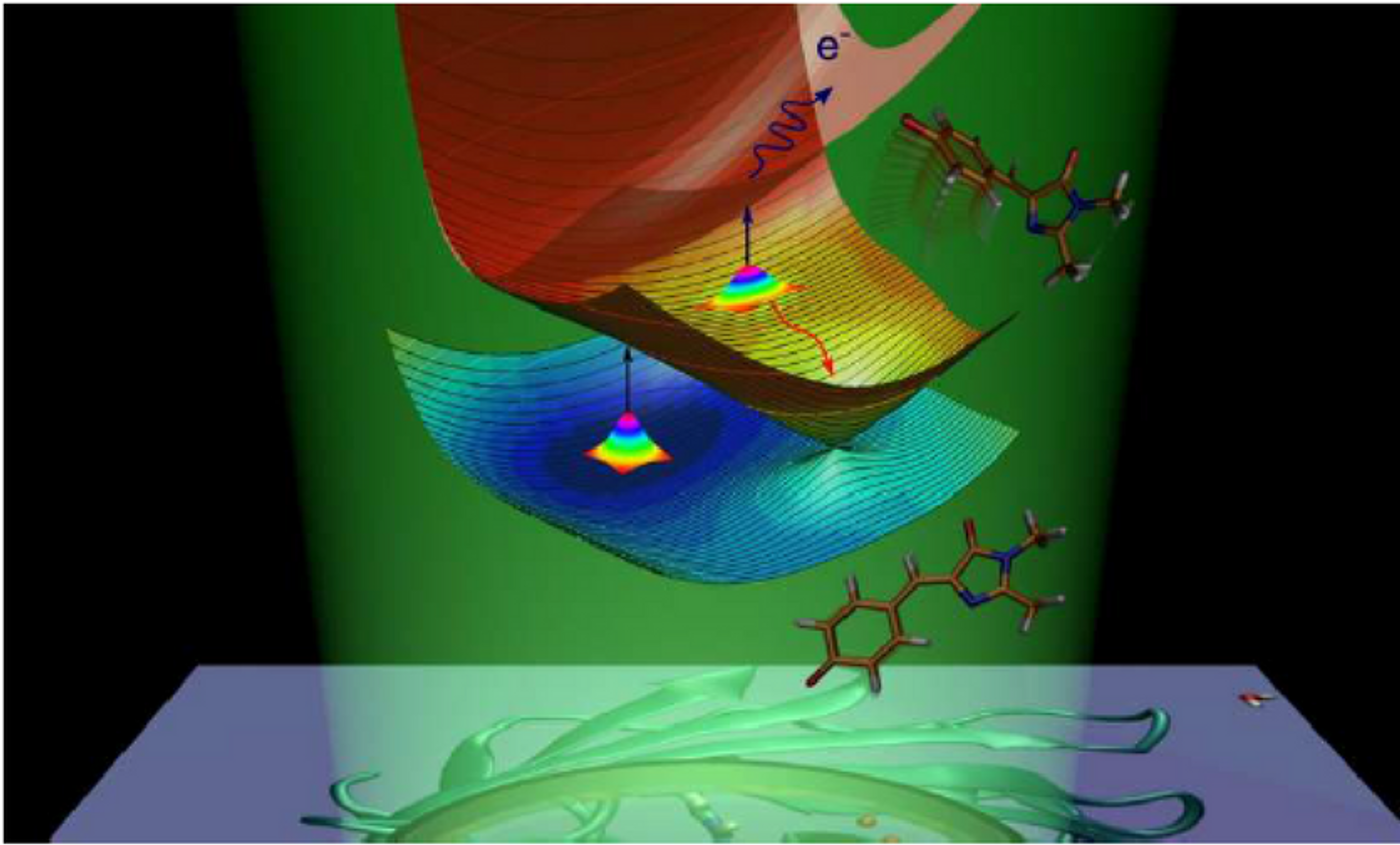
The chromophore of green fluorescent protein. Credit: Anastasia Bochenkova.
In a recent publication of the Journal of the American Chemical Society, a team of Russian and Danish researchers reported that the chromophore of GFP is capable of fluorescing even when it is isolated from its protein environment, and the protein function is to enhance its fluorescent properties.
The story of GFP begins with Nobel Laureate Osamu Shimomura’s research into a glowing jellyfish in the early 1960s. In this fascinating sea creature, he isolated two proteins: one emits blue light, and the other absorbs blue light and emits green fluorescence, which was later known as green fluorescent protein. GFP underwent further studies and development by two prominent scientists (who shared the Nobel Prize with Shimomura) before becoming one of the most widely used tools in modern biology. Martin Chalfie introduced the GFP gene to the genome of C. elegans, which has since enabled the tracking and localization of specific proteins in living organisms; Roger Tsien tweaked the structure of GFP to produce molecules that can emit light of various wavelengths so that labeling complex biological systems with different colors becomes possible.
As a known mechanism, GFP absorbs energy in the form of light and stores it in its chromophore’s electrons, which leads to the so-called excited state. To return to the ground energy state, the protein loses its excess energy through two different deactivation processes: the first one is fluorescence - emitting energy in the form of light, and the second one is called nonradiative relaxation, during which electronic energy, is transformed into vibrational energy of nuclei.
The Russian and Danish scientists discovered that there are intrinsic energy barriers on the nonradiative deactivation pathways, which can trap an isolated GFP chromophore in the excited state. When the chromophore is cooled to 100 K, the barriers enable a slow radiative channel and close the fast nonradiative channel, which causes the isolated molecule to fluoresce. Therefore, the GFP chromophore is intrinsically fluorescent. Interactions between the chromophore and the protein environment allow better entrapment of the electronic energy and keep the chromophore in the excited state. As a result, the radiative channel becomes predominant inside the protein already at room temperature.
"Our research aims at disclosing molecular mechanisms of functioning of living systems. A fundamental challenge is to understand the relationship between the properties of an entire light-sensitive biosystem and its much smaller active chromophore, which is at the heart of the molecular machinery. Here, we have shown that the properties of a single chromophore are important for understanding the mechanisms of action of the entire GFP protein,” explained Anastasia Bochenkova of the Lomonosov Moscow State University and one of the senior authors.
She hopes that beyond solving the question about the origin of the intrinsic fluorescence of GFP, the detailed molecular knowledge of the chromophore might be used together with molecular engineering in designing new functional biomaterials with optimal photo-response.