Better Know a Microbe: Segmented Filamentous Bacteria
Segmented filamentous bacteria (SFBs) are exactly what they sound like - a group of bacteria defined by their morphology. These bacteria were first described in 1974, but they have not been formally classified because they can’t be grown in pure culture (at least not yet). Despite what little we know about SFBs, we do know that they’re important for our immune system (more on that later).
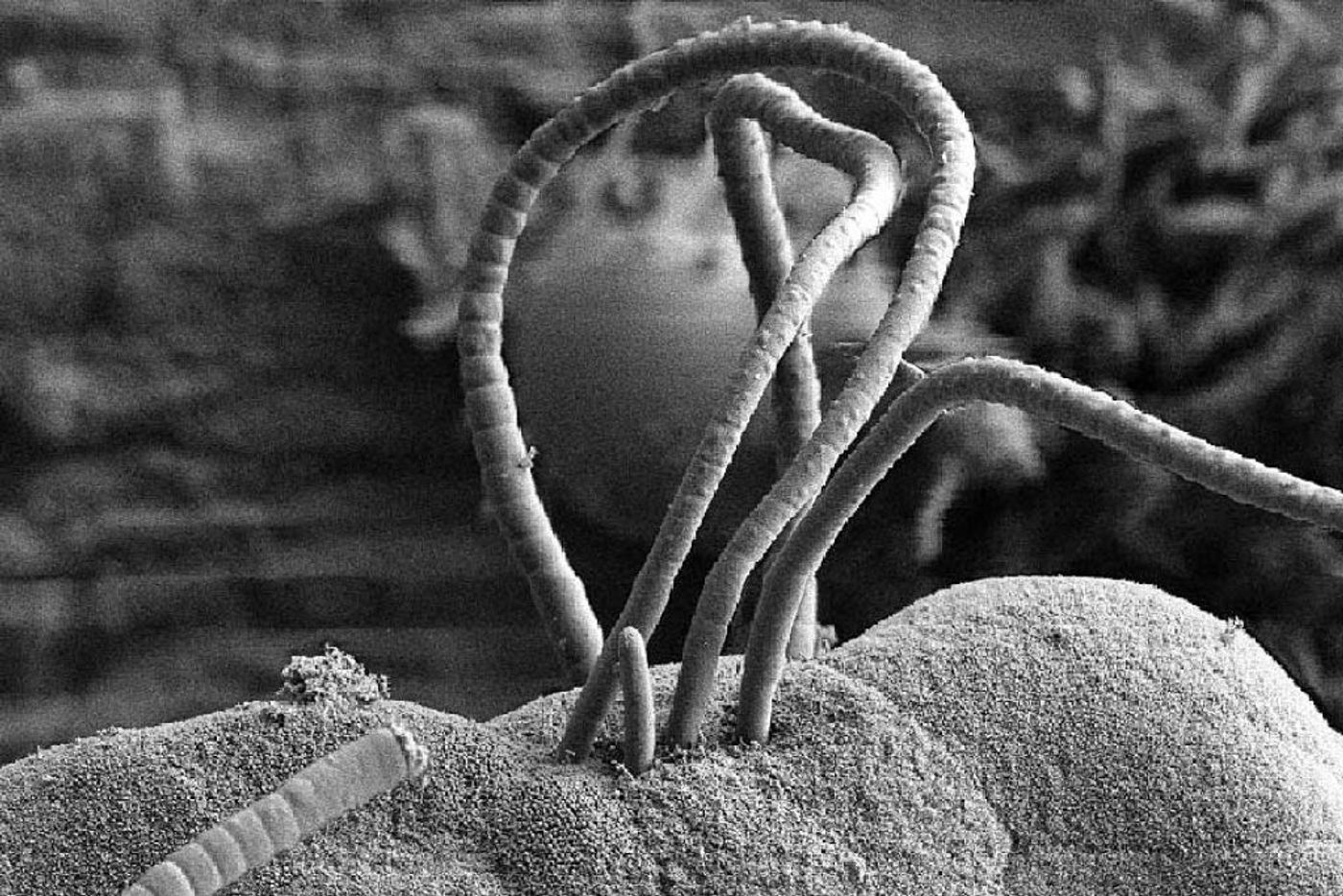
Image: Ivaylo Ivanov, Columbia University
SFBs live in the intestines of many vertebrates, and they colonize some invertebrates as well. The cells grow as long (up to 1 mm!), segmented filaments and were first discovered in laboratory mice and rats. SFBs are Gram-positive, form spores, and don’t cause their hosts any harm - they aren’t pathogens and are considered commensals. Curiously, SFBs anchor themselves to the epithelial cells of the ileum; this association is thought to protect the host from pathogens.
A report from the 1990s examined 14 species of laboratory animals, a small group of humans, and a handful of wild animals for the presence of SFBs. The researchers found SFBs in cats, dogs, rhesus monkeys, crab-eating macaques, domestic fowl, South-African claw-footed toads, carp, rats, and mice. Curiously, not every animal of the same species tested positive for SFBs, and only 1 of the 6 human subjects tested positive.
Although SFBs still can’t be grown in pure culture, a report from 2015 details how SFBs can be grown in a coculture with TC7 cells (a human intestinal epithelial cell line). This report presented evidence to support a hypothesis from the 1970s about how SFBs reproduce. At one end of the SFB, there is a holdfast that attaches to a host epithelial cell. The other end of the cell is called the intracellular offspring tip - as the filament grows and differentiates, intracellular offspring are formed and released from the filament! Pretty wild, if you ask me.
While not formally classified, they are closely related to Clostridia, based on genetic sequencing. One study reported the complete genome sequence of a mouse SFB. (These SFBs were grown in germ-free mice.) Interestingly, the mouse SFB genome lacks a number of genes for the synthesis of amino acids, vitamins, and nucleotides. However, these SFBs do have genes for flagella-based motility and sporulation.
There’s also some convincing data that SFBs bolster the immune system by inducing the differentiation of proinflammatory Th17 cells in the gut. Researchers colonized germ-free and SFB-deficient mice with SFBs. In these experiments, the SFBs induced the differentiation of Th17 cells, without altering the proportions of Th1 cells or Tregs. In addition, compared to germ-free mice, mice that are only colonized with SFBs have more IgA-producing cells in the intestines and higher titers of IgA in the intestines and serum.
For an excellent review of SFBs, I recomment Aaron Ericsson's article in Comparative Medicine.
Sources: Comparative Medicine, Laboratory Animals, Wikipedia, Mucosal Immunology, DNA Research, Taconic, Nature