New Imaging Method Solves Molecular Structures in Motion
Researchers from the State University of New York at Buffalo (UB) announced that they have developed an innovative imaging method to examine biological macromolecules with the finest detail, according to a recently published paper in the journal Nature Method. According to the authors, this revolutionary technique would not only “see inside” of many previously hard-to-define molecular structures, but also can be applied to and transform imaging in various fields, from astronomy to drug discovery.
Scientists nowadays rely heavily on X-ray crystallography to solve molecular structures, a procedure that requires crystallizing the specimen, which is often difficult to achieve, and placing them in non-physiological environments, which could change the conformation of the molecule drastically.
The emergence of cryogenic electron microscopy (Cryo-EM), the technique that earned three of its inventors Chemistry Nobel in 2017, provided much needed help to structural biologists and biochemists who are left down by X-ray crystallography. Cyro-EM allows them to observe specimens that have not been stained or fixed in any way, showing them in their native environment.
"The problem is that more than 75 percent of molecular structures do not readily form the ordered crystals that diffract well. That means many molecules are difficult to visualize in three dimensions," Thomas D. Grant, UB assistant professor and the senior author of the article explained the backdrop of their study.
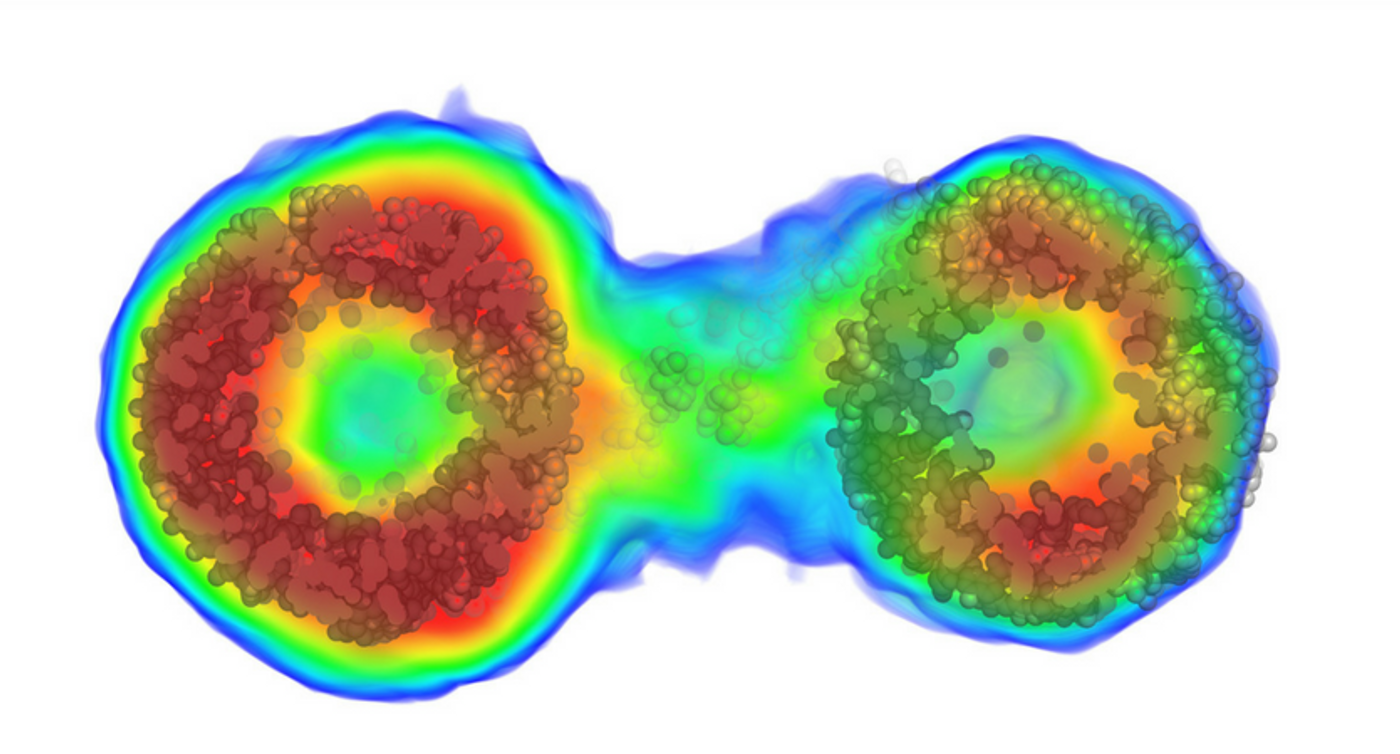
On top of that, biomolecules can exhibit dynamic motions that have an impact on how they function. X-ray crystallography captured stationary images with no movements. Therefore, important biological information could be missing from the picture, which causes the phase problem (critical information about the phase of a molecule is lost during the experimental process of making a physical measurement).
Grant’s team tackled the issues of X-ray-based imaging by solving the phase problem for a particular molecular determination technique called solution scattering. With the improved technique, they can acquired the structures' scatter intense X-rays in patterns consisting of hundreds of thousands of unique pieces of information, which helped to reconstruct molecules with high-resolution.
Then they applied an algorithm called “iterative structure factor retrieval”, which allows them to reconstruct not only the 3-D phases but also the 3-D intensities which are lost in solution scattering experiments as the molecules tumble randomly in solution.
“This is the first demonstration of the ability to reconstruct three-dimensional objects from one-dimensional experimental data, and it will likely have a large impact in related imaging fields,” said Grant.
What is Cryo-Electron Microscopy (Cryo-EM)? Credit: UC San Francisco (UCSF)
Source: UB