Patients can Self-Inject Easily Using an Integrated Pre-Filled Syringe and Autoinjector
The patients suffering from chronic disease require a regular dose of medicines, either orally or as injections. The patients prefer the self-injection method as it is easy to use, cost-effective, and prevents a monthly visit to the specialists.
Because chronic diseases require repeated dosing of medicines, the market for self-injections grew at over 22% per year towards nearly $80 billion by 2022.
“This move towards injection of biologics at home is a clear trend in our society and our industry, and it presents a clear challenge for device developers,” says John A. Merhige, Chief Commercial Officer, Credence MedSystems, Inc.
Video courtesy: Dove Medical Press
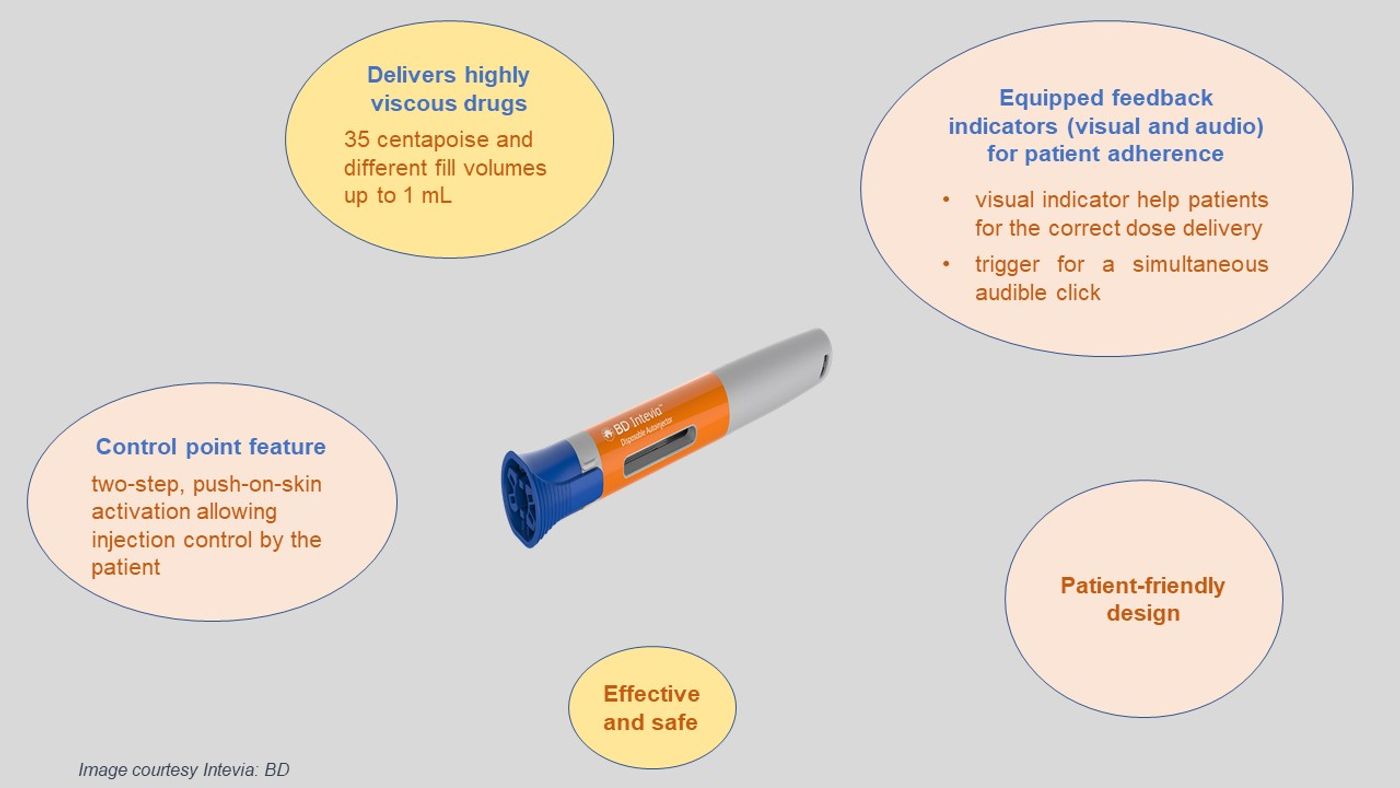
Becton, Dickinson, and Company (BD) is a global leader in medical technology and self-injection systems. BD announced the launch of Intevia at Parental Drug Association’s (PDA) 2019 Universe of Pre-Filled Syringes and Injection Devices conference held in Gothenburg, Sweden. BD Intevia is a 1mL two-step integrated platform device that combines both an autoinjector and a pre-fillable syringe in one system (Image 1 and a Video). The system favorably optimizes patient self-injections with high-viscous drugs and therapies.
Peter Nolan, Worldwide President, BD Medical - Pharmaceutical Systems, said: “BD Intevia is a new generation of BD autoinjectors that will help drug manufacturers seamlessly enhance drug delivery experience.” “For the patient, the platform technology is designed to promote patient ease of use when self-injecting, and it is supported by BD’s worldwide clinical injection expertise harnessing the latest research in drug delivery technology,” he added.
Image 1: The features of BD Intevia
The latest validation study participants highly rated BD Intevia autoinjector-prefill syringe and switching to the integrated disposable system for key product attributes as:
- Overall acceptance, and
- Confidence to control the start of injection.
Source: MarketSearchEngine, BD Intevia