Injury, cell death, and disease to the nerve cells in the eye can cause irreversible vision loss and blindness. But researchers at Johns Hopkins have found a way to grow those retinal nerve cells in the lab that they hope to transplant into patients one day.
The eyes’ nerve cells are called retinal ganglion cells (RGCs). Located in the retina of the eye, RGCs collect visual information from the cones and rods, and then transmit those images to the brain. Our eyes have approximately 0.7 to 1.5 million RGCs, but loss of these nerve cells is irreversible, and could lead to permanent vision loss. Finding a way to make RGCs in the lab could mean a way to restore eyesight.
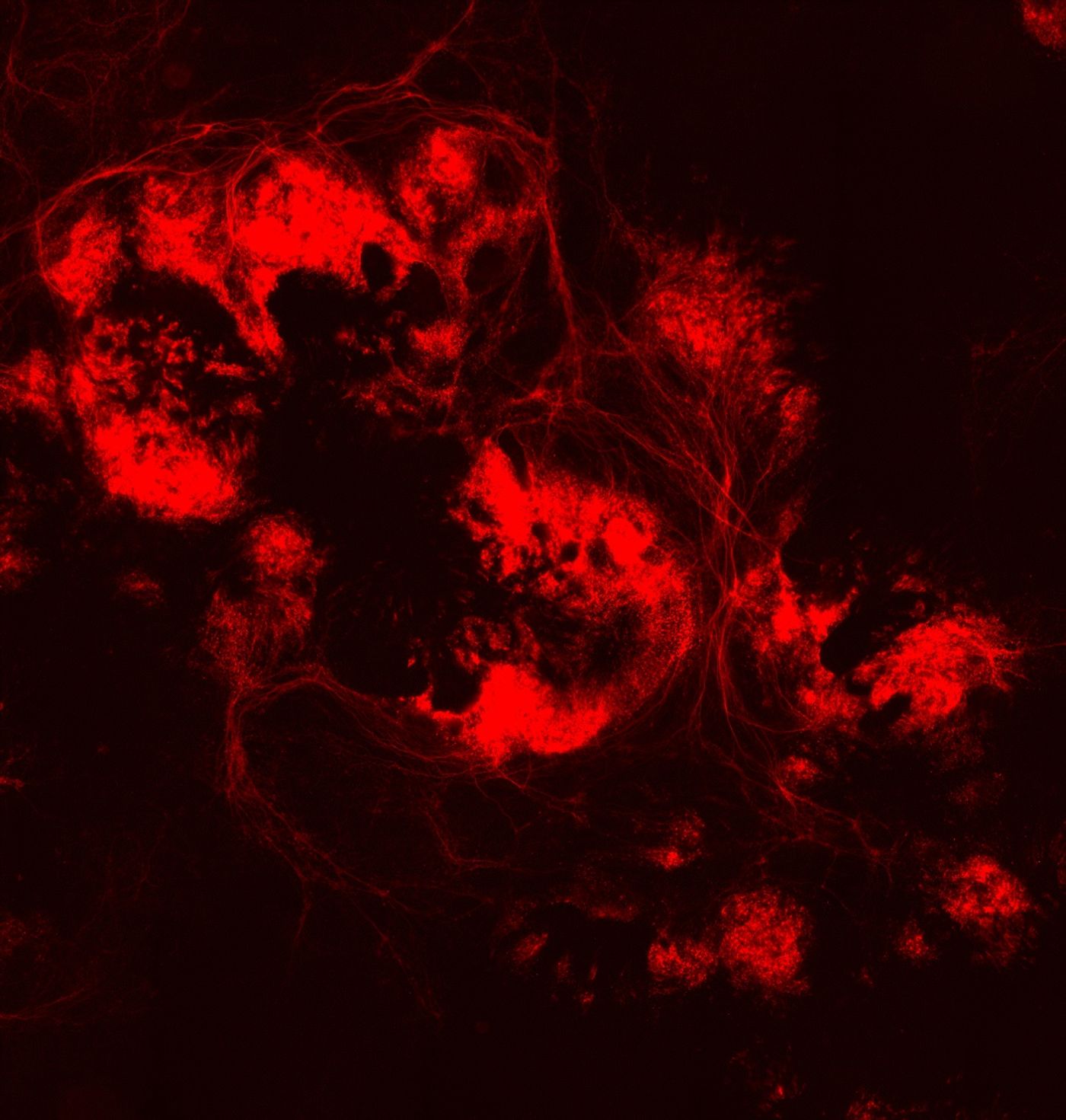
The lab-grown retinal nerve cells began as human embryonic stem cells that have the capacity to differentiate. From there, the researchers used the genome editing CRISPR/Cas9 tool to insert a red fluorescent marker (mCherry) into another gene (BRN3B) that’s expressed only in mature retinal ganglion cells. Researchers found that stimulating the stem cells with a chemical, forskolin, enhanced the differentiation process. Once the stem cells were differentiated into retinal ganglion cells, the fluorescent marker is activated, and the cells glow red.
The red glow of the cells allow researchers to use a method called fluorescent-activated cell sorting (FACS) to isolate differentiated from undifferentiated cells. With continued care by the team, the single red cell grew into clumps of retinal nerve cells. In effect, the red marker allowed them to isolate a highly pure and abundant population of retinal ganglion cells, a first in this type of study.
"By the 30th day of culture, there were obvious clumps of fluorescent cells visible under the microscope. It seems we can now isolate the cells and study them in a pure culture, which is something that wasn't possible before.” Dr. Valentin Sluch, first author of the study.
In addition to looking like the natural version, the lab-grown RGCs also behaved like natural RGCs. They produced similar biological markers, and can fire action potentials like the real deal.
"Our work could lead not only to a better understanding of the biology of the optic nerve, but also to a cell-based human model that could be used to discover drugs that stop or treat blinding conditions. And, eventually it could lead to the development of cell transplant therapies that restore vision in patients with glaucoma and MS." Dr. Donald Zack, senior author of the study.
With the success of growing and isolating pure retinal nerve cells in a petri dish, the team now plans to further tweak the system and adapt the cells for future medical applications. "We really see this as just the beginning. We hope that these cells can eventually lead to new treatments for glaucoma and other forms of optic nerve disease," adds Zack.
Watch the video to learn more about how retinal ganglion cells work in the eye.
Sources:
Nature Scientific Reports,
Hopkins Medicine