Metastasis is one of the biggest challenges facing current available cancer treatments. It is this unpredictable progression of cancer that makes melanoma the most deadly form of skin cancers. But results from a
new study may help doctors track the cancer’s spread by measuring levels of DNA from circulating tumor cells.
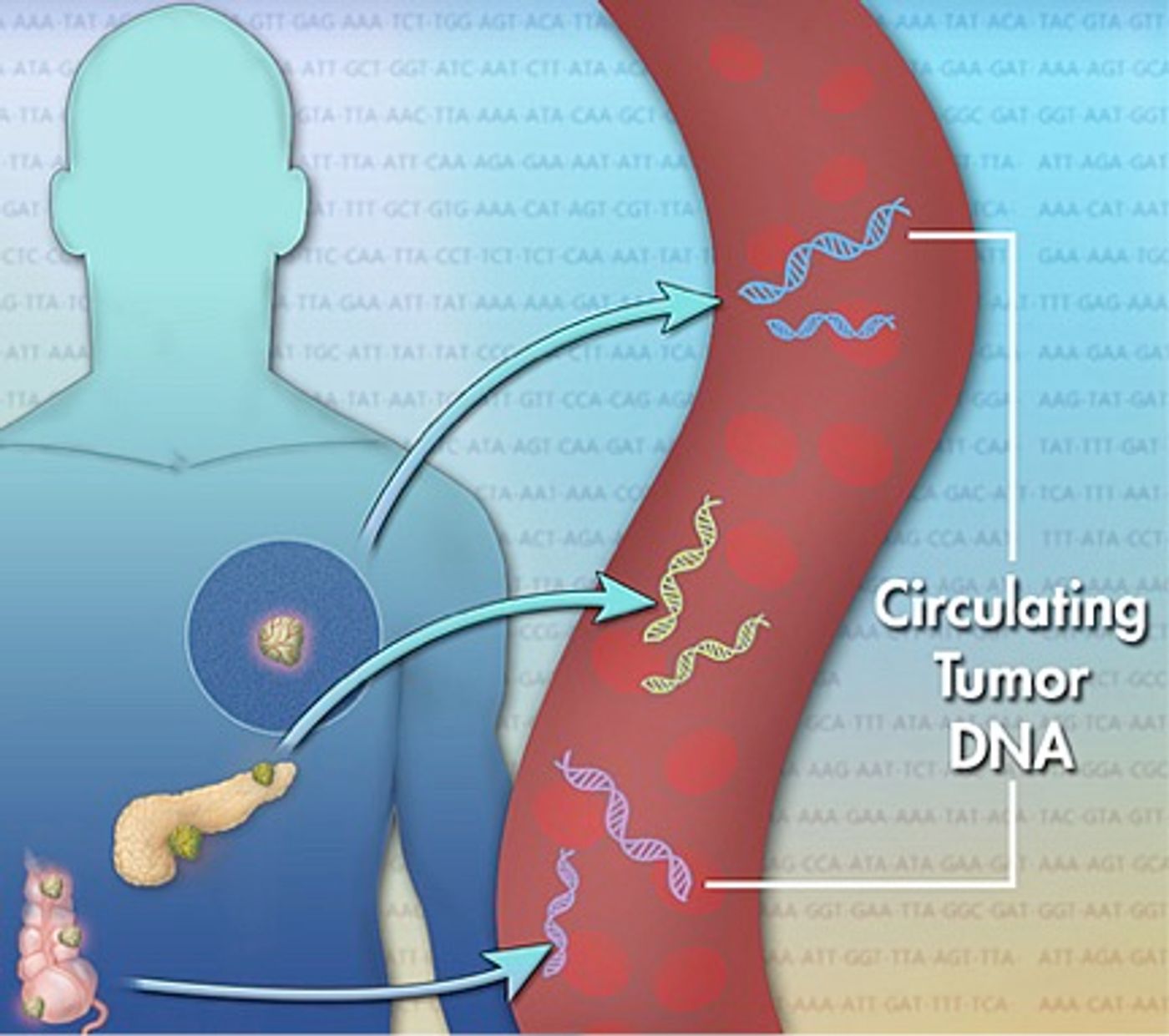
Circulating tumor DNA (ctDNA), also known as cell-free tumor DNA, is genetic material that’s freely floating in the bloodstream of cancer patients. The source of ctDNA is the tumor, where dead tumor cells break open and release the tumor DNA into the patient’s blood. ctDNA represents a non-invasive cancer biomarker that can indicate the presence of disease. And because detection requires only a blood sample, the technique of using ctDNA for cancer diagnosis is often called “
liquid biopsy.”
The researchers proposed that ctDNA could provide an accurate way to track the progression of metastatic melanoma. Since the ctDNA is specific to the melanoma tumor, this new test could provide better diagnostic accuracy than traditional tests that measure an enzyme called lactate dehydrogenase (LDH). Though LDH levels do rise in presence of aggressive tumors, it is also affected by other health conditions, which could convolute the accuracy of the LDH screen.
In 31 patients who had advanced metastatic melanomas, researchers found that ctDNA levels were more predictive of the disease progression than levels of LDH. ctDNA levels were elevated in 80% of the patients, as compared to LDH levels that were elevated in only 30% of patients.
ctDNA levels were also better at predicting cancer recurrence (85% of patients) compared to LDH levels (54% of patients). Recurrence rates were confirmed by X-ray or CT scans.
Overall, the ctDNA screen had higher sensitivity than the LDH screen at detecting the spread of melanoma.
"Our study results show that circulating tumor DNA is a superior blood test for evaluating and tracking progression of metastatic melanoma," said David Polsky, senior investigator from NYU Langone Medical Center in New York City.
While these results are promising, it is important to note that the study was done on a very small number of patients. It will be prudent for the scientists to expand on these preliminary results in a larger cohort of patients and controls. Larger trials should demonstrate with higher confidence the potential use of ctDNA as a biomarker for monitoring melanoma disease activity.
Additional source:
Medicine Net