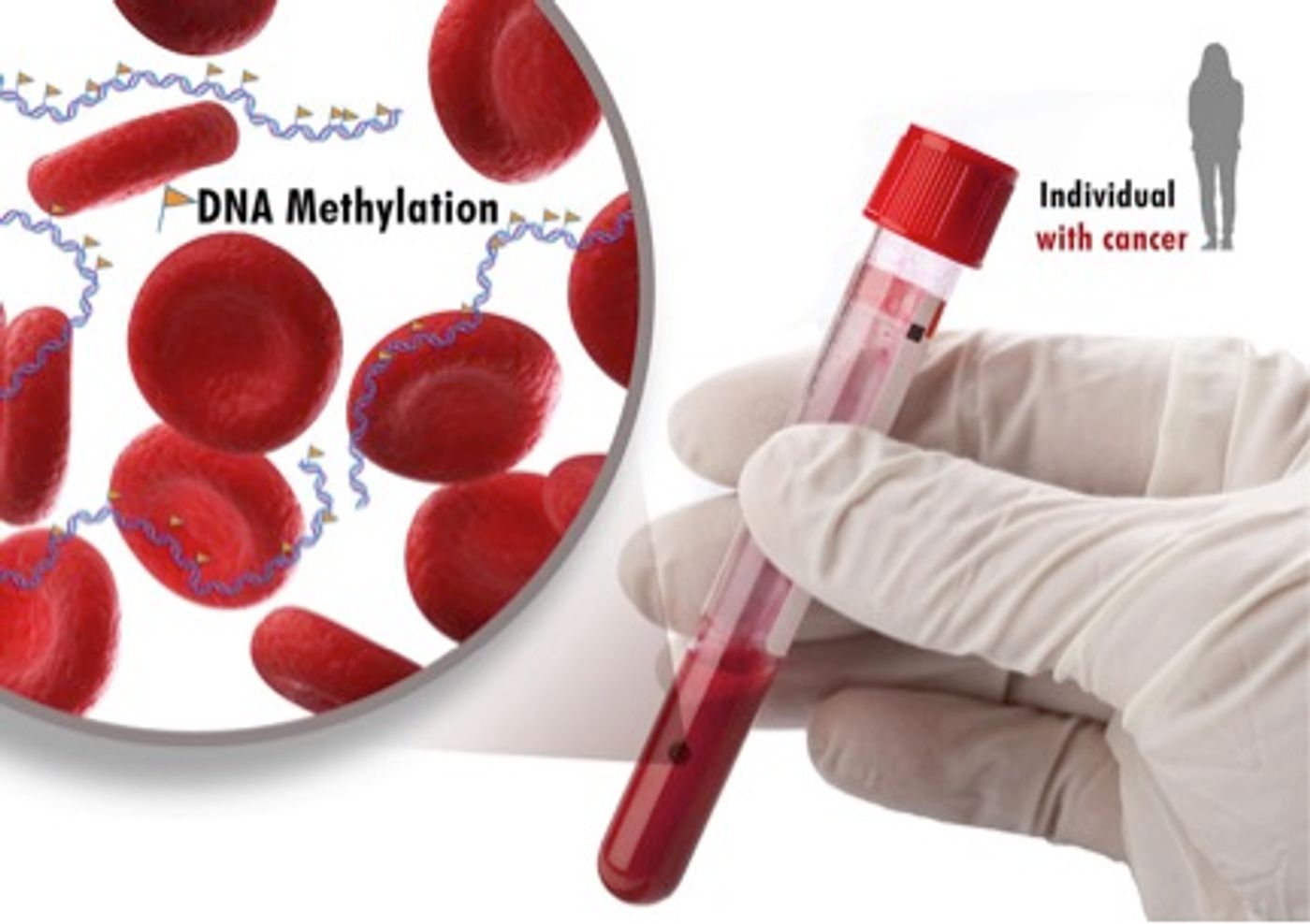
According to researchers from the National Institute of Health (NIH), a simple blood test may soon be able to detect five types of cancer - colon, lung, breast, stomach and endometrial - in the early stages. This diagnostic capability relies on a novel methylation signature present in the tumor DNA.
DNA methylation is an epigenetic process by which chemical methyl groups are added on to DNA bases. Cells use this mechanism to efficiently lock genes in the “off” position and silence gene expression. High methylation (hypermethylation) is correlated with lower gene activity. Conversely, genes that have low methylation signatures (hypomethylated) are more highly expressed.
In 2013, the research team at the NIH’s National Human Genome Research Institute (NHGRI)
originally reported a unique methylation mark around a gene known as ZNF154 that was only present in tumors. The function of ZNF154 is not yet known, though researchers suspect it may play a role in derailing normal cellular processes.
"No one in my group slept the night after that discovery," said Laura Elnitski, Ph.D., a computational biologist and senior leader of the study. "We were so excited when we found this candidate biomarker. It's the first of its kind to apply to so many types of cancer."
Since that publication, the team has worked hard to unmask the same methylation marks in colon, lung, breast, stomach and endometrial cancers. They
now reported that the telltale methylation mark around ZNF154 consistently showed up in all the different tumors types and subtypes. Furthermore, the methylation mark was highly elevated, making detection possible even at low levels.
In the next phase, the researchers will screen blood samples from patients to measure the accuracy of detection from circulating tumor DNA. Computer simulation data with circulating tumor DNA already showed that the abnormal methylation mark could be detected even when researchers diluted 1% tumor DNA with 99% normal DNA materials. They will also have to work out the specificity and sensitivity of the test, assuring that the assay doesn’t miss the cancer or give false positive results.
"Finding the methylation signature was an incredibly arduous and valuable process," said NHGRI Scientific Director Dan Kastner, M.D., Ph.D. "These findings could be an important step in developing a test to identify early cancers through a blood test."
If successful, the test has the potential to diagnose many types of cancer without the need to have prior knowledge of the cancer. This could facilitate detection of cancer at earlier stages, allowing faster and more efficient treatments for the patients. Additionally, the non-invasive test could allow doctors to monitor patients at high risk for developing cancer, or track cancer’s activity during treatment.
"We have laid the groundwork for developing a diagnostic test, which offers the hope of catching cancer earlier and dramatically improving the survival rate of people with many types of cancer," Dr. Elnitski said.
Additional sources:
EurekAlert!