Zika is not going away anytime soon, as cases continue to rise in the Americas. The CDC and the FDA are taking note, and in response to this health crisis, have developed and approved use of an emergency diagnostic test for this disease.
Called the Zika MAC-ELISA, the test detects immunoglobulin M (IgM) antibodies that are present after exposure to the Zika virus. After infection, the body tries to fight the virus by producing the IgM antibodies, which stays in the blood anywhere from 5 days after infection until 12 weeks when symptoms go away.
There are currently no FDA-approved commercial tests for Zika detection, and this new test is issued under the Emergency Use Authorization (EUA). This means that the test will be given under emergency circumstances that impact public health security.
The new diagnostic tool will be critical for evaluating high-risk symptomatic patients. However, there are some caveats to the test results. False positive results may arise if the person was infected with a closely related virus, like the dengue, for example. The test could also give false negatives, as the antibodies don’t show up until several days after the infection, and only last around 12 weeks in the blood. So the timing of the test could influence the results dramatically.
In the next two weeks, the CDC will be distributing the tests to qualified laboratories. Health officials anticipate that the availability of this new test will improve detection and management of the Zika virus in the US, where there are currently 147 confirmed cases of Zika.
The majority of these cases were acquired during travels to Zika-infected areas. Still, Puerto Rico is considered a high-risk area in the US, with CDC Director Tom Frieden confirming that it could have the potential for hundreds of thousands of cases of the virus.
Even more troubling,
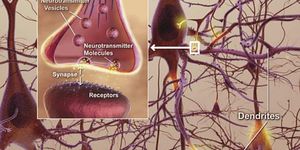
a recent report, published in the medical journal Lancet, found a strong link between the Zika and Guillain-Barre, a rare autoimmune disorder that affects the nervous system. While the incidence of Guillain-Barre is rare on its own, the researchers think Zika may make it less rare.
This new link to Guillain-Barre is not yet firmly established. But add this to the microcephaly link, and the new diagnostic test for Zika couldn’t come sooner.
Additional source:
MNT