Aging May be Reversible in Mice
Growing old has always been a question of not if, but when. This cellular inevitability may not have to be the case, however. A team of researchers from the Salk Institute recently found a molecular way to reverse cellular aging in mice. This discovery has big implications for our youth-obsessed culture. But more importantly, researchers hope the results could someday lead to medical therapies for those who suffer from age-related disorders like progeria.
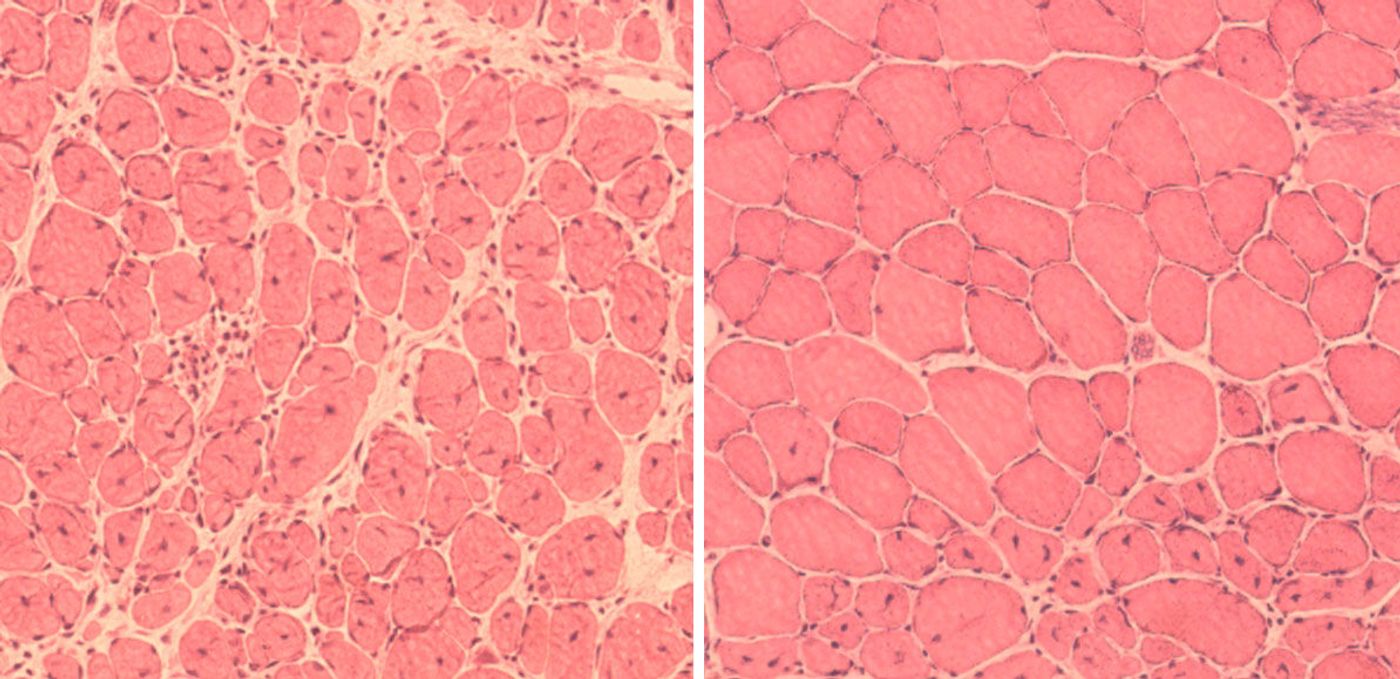
One of the most well-described aging disorder is progeria. Known formally as Hutchinson-Gilford progeria syndrome, this rare genetic condition is often described as accelerated aging. Afflicted children show signs and symptoms of abnormal aging beginning in their first year of life. The skin thins and wrinkles, hair falls out, bones and joints ache, organs fail – the entire system is so fragile that affected children resemble old, aging people. Most progeria patients do not reach adulthood and can barely survive their teenage years.
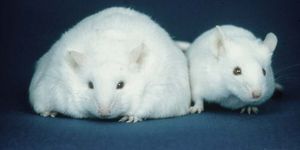
If aging can be accelerated, there must be a way to slow it down. Using a mouse model of progeria, researchers at the Salk Institute’s Gene Expression Laboratory in La Jolla, California experimented with turning on molecular factors that influence cellular aging.
The team focused on the Yamanaka factors - four genes that, if activated, can revert a cell’s state back to an earlier version of itself. These factors have been applied to stem cell research to make induced pluriopotent stem cells, but the technique has not been successfully applied to live animals.
In animals, turning on the Yamanaka factors typically result in the formation of tumors. To bypass this unwanted side effect, researchers created a conditional expression model. Specifically, the Yamanaka factors would only be activated in the progeria mice when they were exposed to an antibiotic called doxycycline.
Incredibly, the short bouts of treatments throughout the animal’s life led to measurable changes in their physical appearance. At the molecular level, the cells appeared more youthful with less damage. On the outward physical level, the mice had less spine curvature and had better functioning organs.
"The mice treated with these reprogramming factors had tissues that were better-looking, they were more healthy and they didn't accumulate the aging hallmarks," said Pradeep Reddy, a research associate at the Salk Institute.
"Our study shows that aging may not have to proceed in one single direction," said Juan Carlos Izpisua Belmonte, the study’s senior author.
Activation of the “reprogramming factors” in aged but healthy mice also resulted in positive effects. Treated mice seemed to have cells that were better at healing and regeneration, effects which surely would help with the aging process.
Although the results are promising, the researchers admit that such response were limited to mouse models, and that results in humans could vary considerably. Still, these results represent an exciting step toward potential therapies for progeria, a condition which does not have any cure in sight.
"Obviously, mice are not humans and we know it will be much more complex to rejuvenate a person," said Izpisua Belmonte. "But this study shows that aging is a very dynamic and plastic process, and therefore will be more amenable to therapeutic interventions than what we previously thought."
Additional sources: Live Science, Salk Institute