Respiratory Syncytial Virus Antigens and Antibodies
Applications in Therapeutic Antibody, Vaccine, and Immunodiagnostic Assay Development
In early April, Pfizer announced a $525 million acquisition of ReViral’s novel therapy for respiratory syncytial virus (RSV). Since then, RSV therapies have garnered renewed global attention. However, although scientists have been studying and developing therapies for RSV infection since the 1960s, a convincing and safe vaccine remains elusive. Thus, this move has sparked some speculation that Pfizer's acquisition could usher in a new era of RSV treatment.
What is RSV?
RSV is the leading cause of lower respiratory tract infections in infants and children worldwide, with high morbidity and mortality rates. In the elderly, RSV reinfections result in fever, pneumonia, and even death. According to the Centers for Disease Control and Prevention, each year in the USA alone, RSV causes 58,000 hospitalizations of children under the age of 5 years and 177,000 hospitalizations of older adults aged 65 years or more. Furthermore, of the RSV infection cases in older adults, 13,000 were fatal.
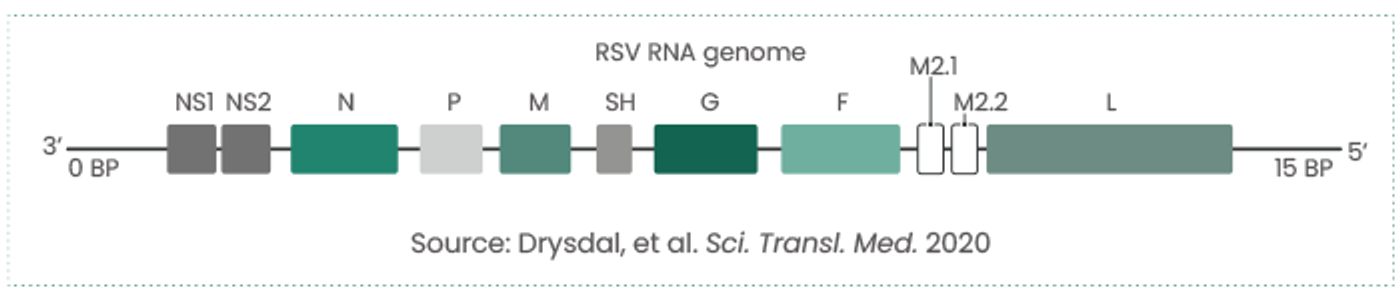
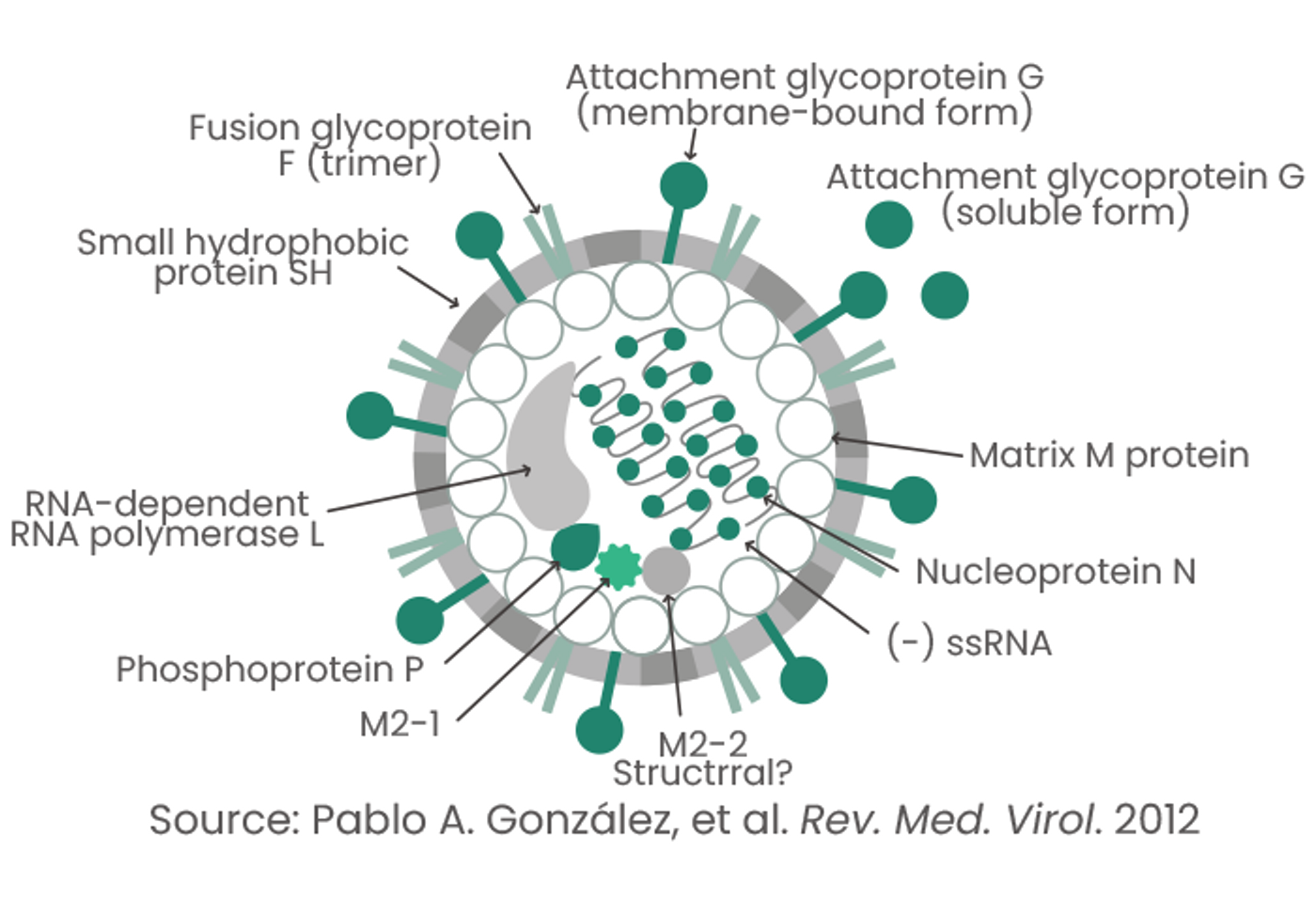
RSV is an enveloped negative-strand RNA virus belonging to the family Paramyxoviridae. It contains 10 genes that encode 11 proteins.
RSV has two major subtypes: A and B. Although both strains often cocirculate, only one predominantly causes infections in any specified period.
RSVs typically comprise several envelope and structural and nonstructural proteins:
- Envelope proteins: G glycoprotein, fusion (F) glycoprotein, and small hydrophobic (SH) protein
- Structural proteins: Large (L) protein, nucleocapsid (N), phosphoprotein (P), matrix (M), M2-1, and M2-2
- Nonstructural proteins: NS1, NS2
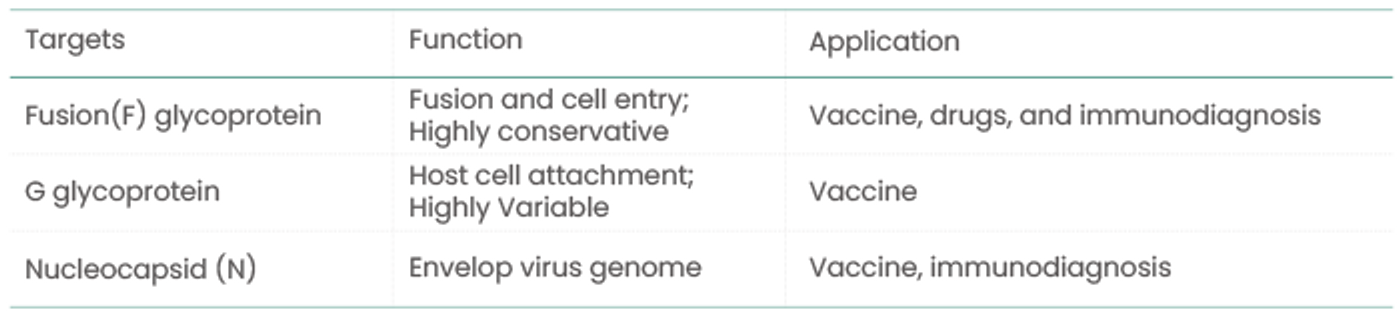
The following are the most frequently developed targets for vaccine, drug, and immunodiagnostic development:
How can RSV Reagents be Applied in Research?
Vaccine Development
Although over 60 RSV vaccine programs are currently in development worldwide, no vaccine has been approved for clinical use. Throughout the development process, scientists have been able to employ extensive knowledge of viral vaccines. For example, most vaccines target the fusion (F) glycoprotein. However, G glycoprotein is less frequently used in RSV vaccine research due to its highly variable characteristics. Sino Biological’s RSV reagents have aided in preclinical studies, clinical trials, and quality control sessions during the development of the RSV vaccine, including vaccine content testing, vaccine efficacy testing, and toxicology research.
In a previous study of DNA vaccine delivery mechanisms, Smith et al. (2017) used recombinant RSV-F protein (Cat#: 11049-V08B) to detect serum antibody titers following vaccination in his study of the DNA vaccine delivery mechanism. The data summarizes the findings of a preclinical study examining the efficacy and safety of a vaccine delivery system (Smith, et al. Vaccine. 2017). Similarly, another study used recombinant RSV-G protein (Cat#: 40830-V08H) to detect RSV A2 virus-specific antibodies in serum.
Therapeutic Drug Development
AstraZeneca’s Synagis® (Palivizumab) is the only globally approved drug for the prevention of RSV infection. It is a humanized murine monoclonal antibody that inhibits the spread of the virus to the lower respiratory tract via deactivation of the RSV fusion glycoprotein. It is indicated only for high-risk preterm infants and not for postinfection treatment. Numerous monoclonal antibodies and small molecule drugs are still being investigated to develop treatments for RSV infection.
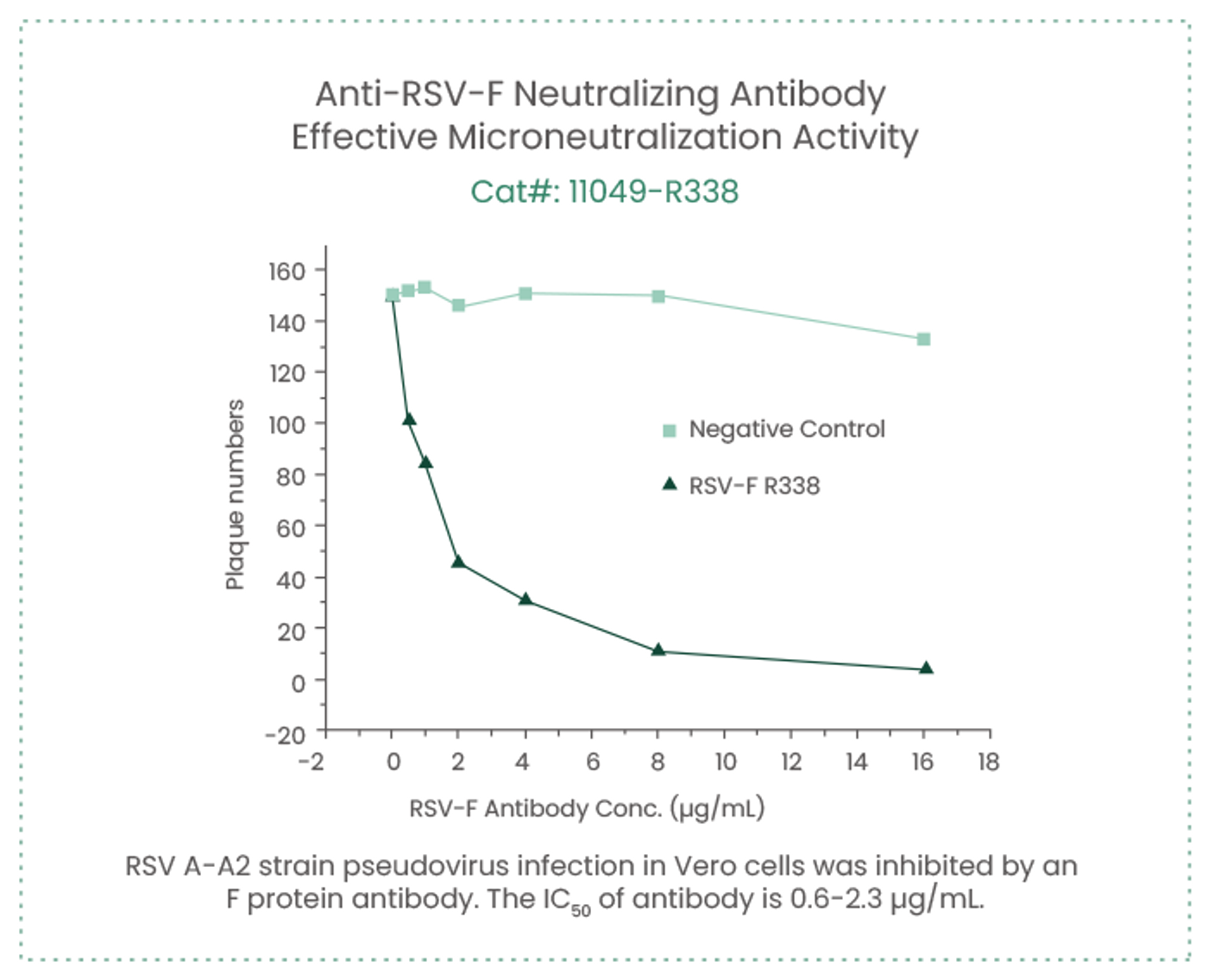
In a previous study focusing on the production of immunoglobulins (Igs) that met the specific therapeutic needs in the treatment of infectious, Jacque et al. prepared affinity chromatography media for anti-RSV immunoglobulin enrichment using recombinant RSV F protein (Cat#: 11049-V08B); they used a rabbit monoclonal antibody (Cat#: 11049-R009) in the antiviral assay to detect the level of F antigen expressed by RSV infected cells following incubation with anti-RSV Ig. The level of F antigen expression reflected the neutralizing efficiency of anti-RSV Ig. (Jacque et al. Front. Immunol. 2021)
Development of Immunodiagnostics
Antigen testing is convenient and rapid, and its sensitivity and specificity are increasing as antigen detection technology advances.
The SofiaTM assay platform developed by Quidel was reported to have a sensitivity and specificity of 78.6% and 93.9 %, respectively, while the VeritorTM assay system developed by BD had a sensitivity and specificity of 81.6 % and 99.1 %, respectively (Cameron Griffiths et al. Clin. Microbiol. Rev. 2017).
Sino Biological’s recombinant RSV N and RSV-F proteins are currently used to prepare and screen core raw materials (antibody pairs) for RSV antigen detection kits, as well as a quality control sample. The company has screened a pair of antibodies that recognize fusion glycoprotein from a variety of RSV strains and can be used as raw materials for RSV detection (Capture antibody, Cat#: 11049-R338; Detection antibody, Cat#: 11049-R302).
Reagent Toolkit for RSV
Sino Biological has developed a comprehensive collection of RSV-related reagents for viral research (17 antigens, 7 antibodies, 80+ ELISA Kits, and cDNAs) that cover RSV A and B from over 5 strains.
Recombinant RSV Antigens
Anti-RSV Antibodies
Anti-RSV-F Rabbit Monoclonal Antibody
For more RSV related reagents, please visit:
https://www.sinobiological.com/research/virus/respiratory-syncytial-virus