Further Challenges in Alzheimer Drug Development

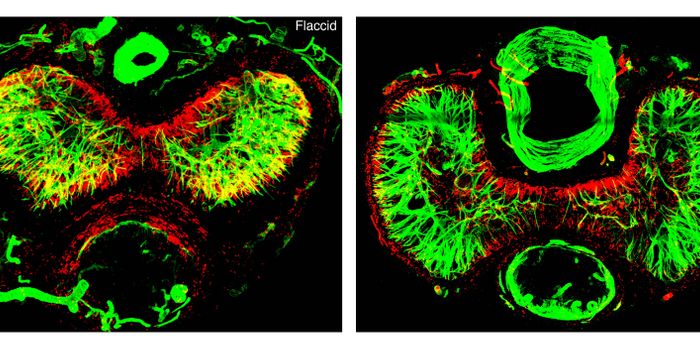
Scientists at Trinity College Dublin performed a large-scale international study involving the treatment of Alzheimer's disease, which was published in PLOS Medicine. The research involved a clinical trial testing a new treatment against a placebo. The study involved a clinical trial, known as NILVAD, which tested 511 participants with mild to moderate Alzheimer’s disease. The clinical trial was testing a new treatment, a medication called Nilvadipine, against a placebo.
The drug, Nilvadipine, is approved to treat high blood pressure. However, in animal studies, it was found to reduce brain amyloid, the toxic protein associated with Alzheimer's disease. Thus, Nilvadipine was seen as a potential treatment for Alzheimer’s. It was therefore considered to be a possible effective treatment for Alzheimer's disease in patients.
The most common form of dementia, Alzheimer's disease affects approximately 34 million people worldwide and as of now, there is no treatment that can delay the onset or slow the progression of the disease.
Results of the study showed that although Nilvadipine is tolerable in individuals with Alzheimer's disease. No effects on memory and function were evident from the drug treatment. "The outcome of the trial for the overall combined group of people with mild and moderate Alzheimer's disease was negative; however, when we broke it down according to severity, those with mild disease appeared to benefit from Nilvadipine whereas those with moderate disease seemed to do worse on the medication. These findings will need to be followed up and teased apart in future studies. It may be that to be successful, we would need to target people at the earliest phase of the disease process,” explains lead author of the study, Professor of Brian Lawlor.
Findings from the paper further implicate drug development for Alzheimer’s.
Source: PLOS Medicine
-
MAY 07, 2024Is It Anti-RNP or Anti-Sm/RNP?
-
MAY 08, 2024Expand your Multiomic Capabilities with RNAscope™
- See More
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
MAY 23, 2024For the Love of Digital PCR 2024
- See More