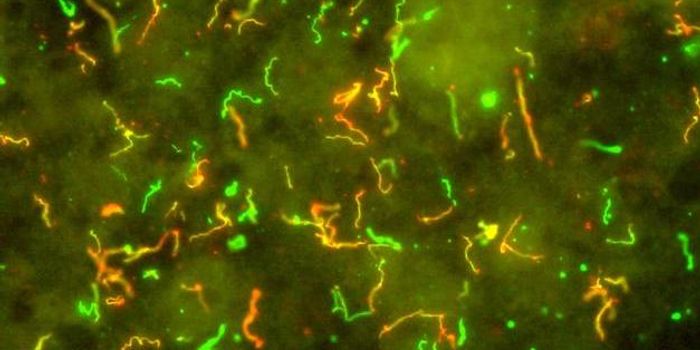
For decades, anti-malaria drugs--known as Chloroquines, have used to treat cancer. But the role in repurposing these drugs for slowing tumor growth have never been quite understood until a recent research study in Cancer Discovery.

The research, carried at out Abramson Cancer Center of the University of Pennsylvania, identified the chloroquines specific target in cancer – an enzyme called PPT1. Identifying that particular enzyme has opened the doors for potential cancer treatments. “The discovery of this target is critical because chloroquines are currently being evaluated in clinical trials all over the world, including here at Penn, and this knowledge fundamentally changes the way we look at those trials,” says study co-senior author and associate professor--Dr Ravi K. Amaravadi of the Perelman School of Medicine at the University of Pennsylvania.
The enzyme, PPT1, controls the target of rapamycin (mTOR)—which is highly involved in the growth of cancer cells as well as another cellular process known as autophagy—a way for cancer cells to detoxify and repair themselves. The mechanisms of autophagy was discovered by Nobel Prize winner-- Yoshinori Ohsumi, watch the video below to learn more:
In previous studies, autophagy was found to provide nutrient support for mTOR to direct cancer growth while, interestingly, mTOR was found to shut down autophagy when nutrients aren’t needed. Using these studies, the researchers utilized CRISPR/Cas9 gene editing approach to knockout PPT1 from cancer cells and observe if the same effects in earlier research can be seen. Results showed that indeed removing ppt1 does slow tumor growth. “The edited cells look like they’ve been treated with a drug, and they grow significantly slower than the unedited cells,” Prof Amaravadi said. “We also compiled data from existing databases and found PPT1 is both highly expressed in most cancers and also associated with poor outcomes.”
These findings encouraged researchers to develop potent chloroquine known as DC661, that will take advantage of this novel treatment pathway.
“We now have a specific molecular target in cancer, as well as a potent way to reach it,” Prof Amaravadi said. “It not only provides new context for current clinical trials involving hydroxychloroquine, but also, with further development of these compounds toward clinical drug candidates, it opens the door for head-to-head testing of our compounds or their optimized derivatives versus current chloroquines to see which is more effective.”
Source: Drug Target Review
-
MAY 07, 2024Is It Anti-RNP or Anti-Sm/RNP?
-
MAY 08, 2024Expand your Multiomic Capabilities with RNAscope™
- See More
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
MAY 23, 2024For the Love of Digital PCR 2024
- See More