Insight Into the Molecular Basis of Obesity
Obesity is a major health problem around the world, impacting adults and an estimated 41 million children. Researchers have long been trying to learn more about the causes of obesity, and now a collaborative effort has revealed genes that influence the development of regions in the brain that are involved in body weight regulation. Reporting in Cell, teams led by Sadaf Farooqi, Ph.D., FRCP, FMedSci, of the University of Cambridge and Children's Hospital Los Angeles' Sebastien Bouret, Ph.D. have identified a signaling molecule that is critical to the maintenance of a healthy body weight.
"We know that the brain, in particular an area called the hypothalamus, has a very important role in the regulation of food intake and blood sugar," explained Bouret, who is an associate professor of pediatrics at the Keck School of Medicine of USC.
The hypothalamus is known to house neuronal cells that help control appetite and satiety, making it a focus of obesity research. "What we don't yet understand, is how these circuits in the hypothalamus are being organized,” said Bouret. “We want to know how the brain puts itself together and what exactly governs that process." If there is dysfunction in those neuronal circuits, it may be playing a role in obesity. Understanding that dysfunction, which may begin at the earliest stages of brain development, may help reduce obesity rates.
In the Bouret lab, researchers are trying to learn more about how and why certain brain cells connect, and others avoid one another. These intricate connections help the hypothalamus maintain careful control of hunger and satiation.
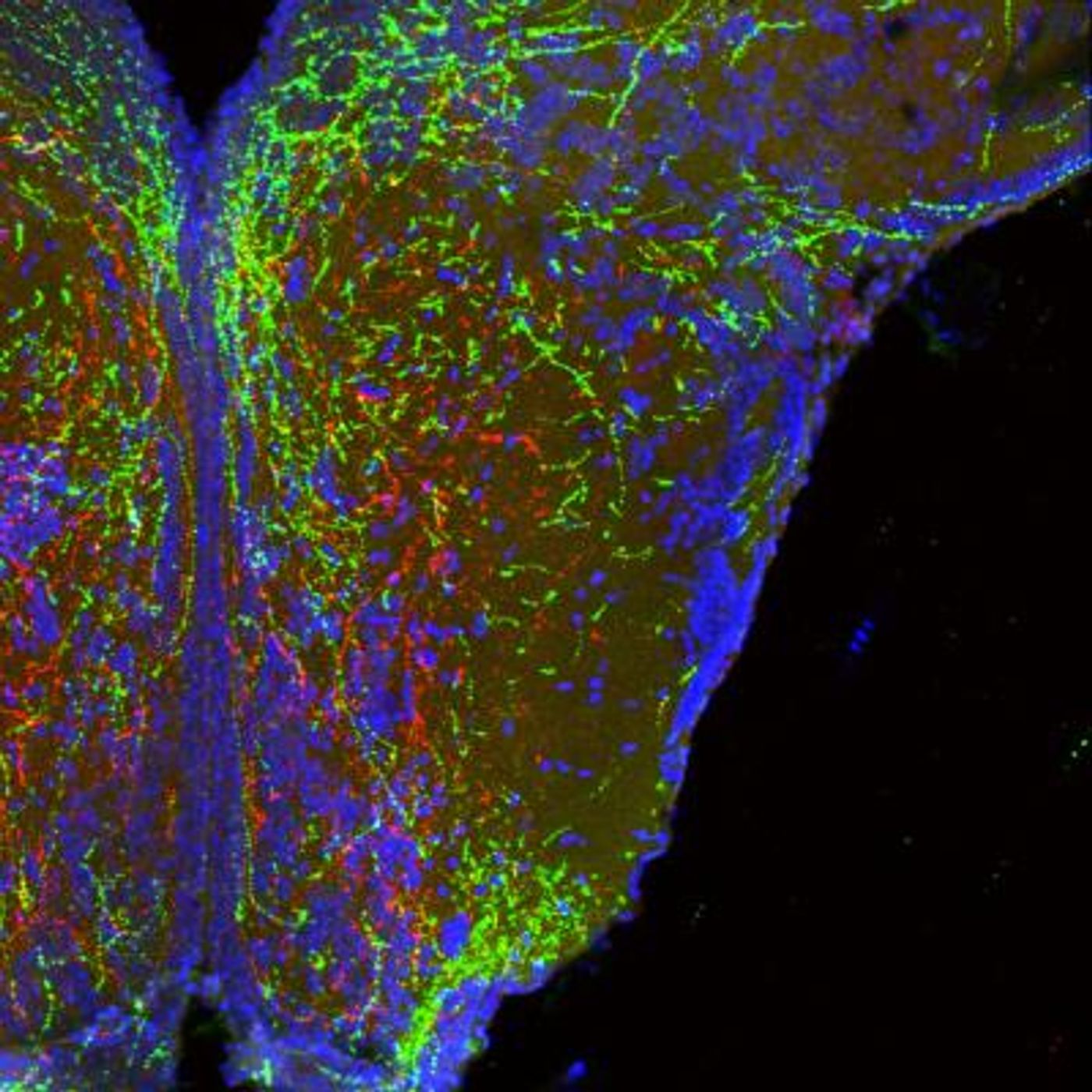
One group of molecules, semaphorins, are plentiful in the developing hypothalamus, and are released by brain cells as signals; they help certain cells reach one another and stay away from others. Using an animal model, Dr. Sophie Croizier of the Bouret lab found that when semaphorin signaling is lost in the hypothalamus, cells there stopped growing the way they should. When that happened, certain connections were lost, and in those animals, body weight increased.
"What we are seeing is that semaphorins are guiding and shaping [the] development of hypothalamic circuits that ultimately regulate calorie intake," noted Bouret.
At the University of Cambridge, a team led by Professor Farooqi assessed genetic data from 1,000 human samples. They found that people with early-onset obesity carried more rare mutations in genes that played a role in semaphorin signaling compared to individuals of normal weight.
That seems to confirm that semaphorin signaling is critical to the maintenance of a healthy weight.
"We have now discovered the genes that establish the precise neural connections that form these circuits," said the co-first author of the study Dr. Agatha van der Klaauw of the Farooqi lab. "This work provides new insights into the development of hypothalamic circuits that regulate appetite and metabolism."
In the video above, Dr. Farooqi discusses genes that play a role in body weight.
Sources: AAAS/Eurekalert! Via Children’s Hospital Los Angeles, WHO, Cell