Mutation Hotspot in Common Fungus Gives it Adaptation Skills
About half of all adults carry the fungus Candida albicans in their gastrointestinal tracts, which probably doesn’t have much of an effect on their health. The fungus can cause a serious and even fatal blood infection, however, in people with compromised immune systems. Researchers at Brown University have now determined how the fungus is able to live in these very different environments. They have also found that in patients, the fungus often lacks one copy of a regulatory gene, which seems to allow the microbe to adapt. The work has been reported in Cell Host & Microbe.
"Diversity in a microbial species protects them against changes in their environment. If they have diversity, a subpopulation of cells may be able to adapt, even if the rest of the population cannot. We found that some samples of Candida albicans can switch phenotype because they're already carrying one inactivated copy of a gene in this genetic hotspot," explained Richard Bennett, a professor in Brown's Department of Molecular Microbiology and Immunology.
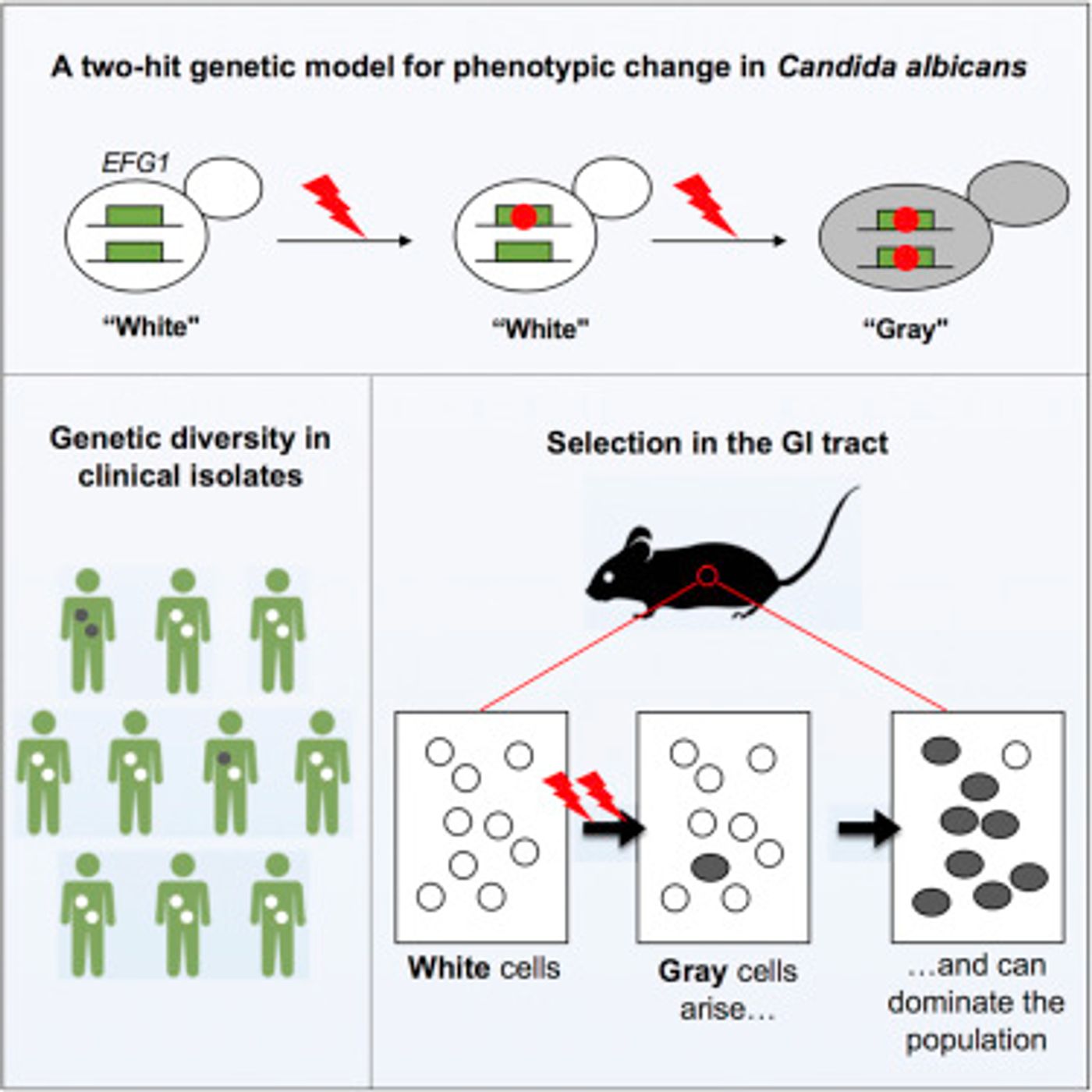
When yeast cells in the lab lose one copy of the EFG1 gene (a master transcription factor), they exhibit different traits. Cells with one or two copies of the gene are white, and in blood, produce virulent infections. Cells without both copies of EFG1 are gray, and are found mostly in the gut.
Bacteria are known to use genetic hotspots, but it was somewhat surprising to find them in yeast, Bennett said. That hotspot promotes diversity that will allow the yeast to thrive in a variety of environments.
Candida, for example, seems to inhabit the gastrointestinal tract of many adults without impact. But it can also cause oral thrush and yeast infections, and in people with a weak immune system, Candida can infect the blood, which is fatal about half the time. That’s because it’s difficult to treat, Bennett said.
In this work, the researchers assessed 63 samples from patients with bloodstream infections, oral thrush, and gut infections; seven didn’t have one or both copies of EFG1. The EFG1 gene produces a protein that controls characteristics like virulence and metabolism. EFG1 plays a role in the virulence of Candida. Two samples that lacked both copies of the gene were found - one in oral thrush and one in a blood infection. The researchers identified numerous repetitive regions of DNA, suggesting to Bennett that a mutation hotspot is at work.
"The EFG1 gene seems to central to a lot of Candida albicans biology, so it keeps getting hit by mutations again and again," Bennett said. "It is interesting that these repetitive sequences can be mutated very easily, and losing this gene is clearly providing some type of selective advantage."
Mutations aren’t always harmful (as discussed in the video). To assess how Candida strains performed when they were competing, the researchers mixed equal amounts of a strain with both copies of EFG1 (white cells) with a strain lacking one copy (gray cells). After adding the mixture to the guts of mice and analyzing what was present over several weeks, the researchers found that cells lacking only one copy were losing their second copy; the gray cells (lacking both copies) began to dominate the cell population.
The researchers want to continue this work by tracking patients over time - obtaining samples from a variety of places in the body and observing how Candida changes within patients. This work can help show how EFG1 mutations influence how people respond to Candida, and how different types of fungi react to treatment. Bennett wants to know more about the mutation hotspot, including how frequently mutations happen there compared to other places in the genome.
"A big question for all microbiologists is: 'How representative is taking a single isolate, or a single cell, of the species overall?'" Bennett noted. "We've learned that while studying one isolate is a good place to start, it's not the whole picture, particularly when you're talking about microbes."
Sources: AAAS/Eurekalert! via Brown University, Cell Host & Microbe


![WGS for rare disease diagnosis [eBook]](https://d3bkbkx82g74b8.cloudfront.net/eyJidWNrZXQiOiJsYWJyb290cy1pbWFnZXMiLCJrZXkiOiJjb250ZW50X2FydGljbGVfcHJvZmlsZV9pbWFnZV84MmRlM2UyYjA5M2Q3ZTYwOTI3Zjc1YTRjOWU2N2RmMjkzMThjMTJkXzI1MDcucG5nIiwiZWRpdHMiOnsidG9Gb3JtYXQiOiJqcGciLCJyZXNpemUiOnsid2lkdGgiOjcwMCwiaGVpZ2h0IjozNTAsImZpdCI6ImNvdmVyIiwicG9zaXRpb24iOiJjZW50ZXIiLCJiYWNrZ3JvdW5kIjoiI2ZmZiJ9LCJmbGF0dGVuIjp7ImJhY2tncm91bmQiOiIjZmZmIn19fQ==)