Making Bad Fat Turn Good
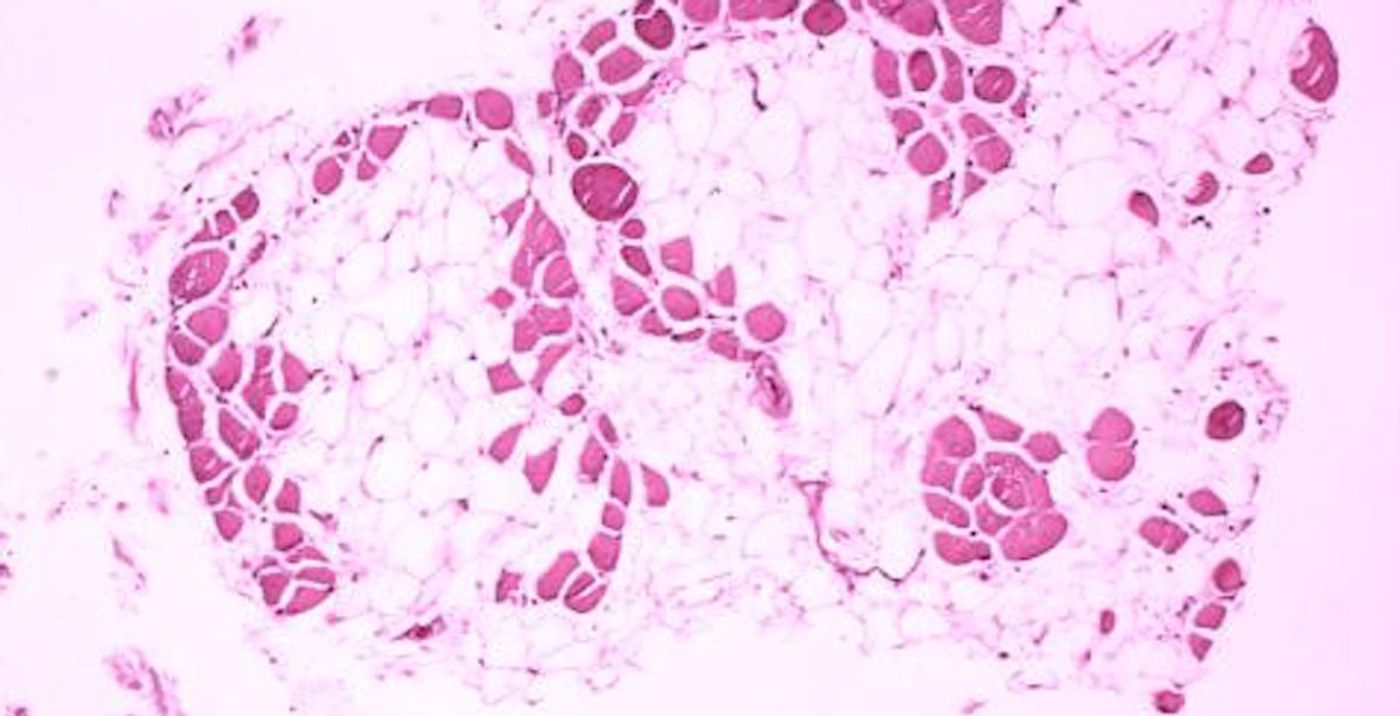
Not all fat cells, also called adipocytes, are the same. White fat or adipose tissue is mostly used as a place to store extra calories and contains large lipid droplets. Brown fat or adipose tissue, on the other hand, doesn’t carry large lipid droplets and instead is rich in energy-generating organelles called mitochondria. Brown fat can generate heat, so it burns energy rather than store it. Fat cells can also release hormones and play an important role in controlling the balance of energy in the body.
"Adipocytes are not merely an energy storage for times of deprivation, but they also release hormones into the blood, regulating our metabolism as well as feelings of hunger and satiety through the brain and other organs. Nevertheless, too much of a good thing causes harm." explained Professor Klingenspor, Chair of Molecular Nutritional Science at the Technical University of Munich (TUM) Else Kröner-Fresenius Center.
Infants carry a lot of brown fat; it helps keep their bodies warm, especially since they don’t shiver. The release of heat from fat cells (to stabilize body temperature) is called non-shivering thermogenesis.
Adults carry brown fat cells too, but the level can vary from person to person. It has been suggested that people with high levels of thermogenic adipocytes have a lower chance of developing metabolic disorders like obesity. Researchers are interested in learning more about how white fat can be converted to healthier beige or brown fat; fat cells that serve as an energy-storing organ could be transformed into an organ that releases energy and improves metabolic health. However, there’s still a lot we don’t know about how this change happens.
"We want to understand how thermogenic fat cells develop; so how beige fat cells grow inside white fat tissue," said Klingenspor.
Different mouse strains are known to have varied abilities when it comes to browning white fat, and that gave the researchers some insight into the process. "By systematically comparing fat cells among these different strains of mice, we were able to discover which genes or regulators might explain the variation in beige cell differentiation - in other words, the growth of beige fat cells," said Klingenspor.
In new work published in Cell Reports, a team of researchers including Klingenspor looked at all the genes that were being expressed, called the transcriptome, in fat cells from different strains of mice. They identified genes that play a role in turning white fat to beige, as well as placing those genes within the context of a gene network.
"Now we have gathered a unique insight into the genetic architecture driving the molecular mechanisms of beige fat cell development. What we managed to confirm in cell culture is now to be examined in vivo; so inside a living organism, as our next step," said Klingenspor.
Sources: AAAS/Eurekalert! via Technical University of Munich, Cell Reports