'Chromosome Shattering' is Common Across Cancer Types
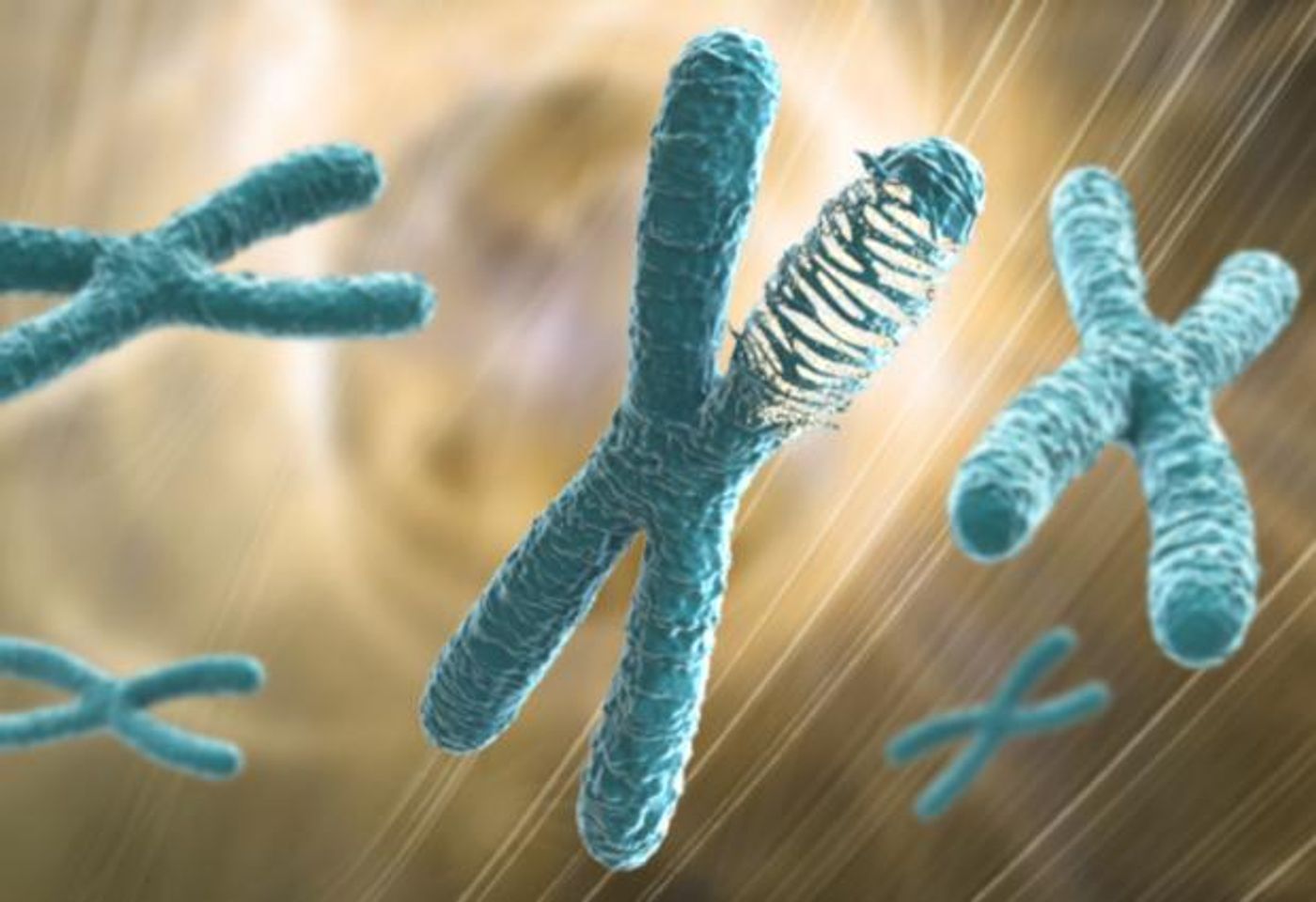
In recent years, scientists have begun to learn more about a type of mutation called chromothripsis that was discovered a few years ago in chronic lymphocytic leukemia. The term chromothripsis literally means chromosome shattering; a catastrophic rearrangement of the genome occurs when chromosomes break into many pieces, then natural DNA repair processes try to fix the damage by stitching the pieces back together. Now researchers have investigated the occurrence of this mutation in different kinds of cancers, as one project in the Pan-Cancer Analysis of Whole Genomes (PCAWG), a massive international collaboration involving more than 1300 researchers.
This work was reported in Nature Genetics, by a team of scientists from Harvard Medical School and European Molecular Biology Laboratory - European Bioinformatics Institute EMBL-EBI). They used whole-genome sequencing (WGS) data from 2,600 tumors representing 38 types of cancer. They found that chromothripsis was relatively common in many kinds of cancer, far more than was thought. Common characteristics of the cancer genome were easy to connect to chromothripsis directly, like an increased number of genes that drive cancer, called oncogene amplification, or a reduced number of tumor suppressing genes, which control the growth and division of cells.
"We integrated WGS data from over 2,600 tumors spanning more than 30 cancer types," said Isidro Cortés-Ciriano, Group Leader at EMBL-EBI. "From this, we discovered that chromothripsis events and other types of complex genome rearrangements are pervasive across human cancers, with frequencies greater than 50 percent of tumors in some cancer types."
Previous work that analyzed the cancer genome has often used genetic tools that are based on arrays, and have low resolution. This work utilized WGS data for a better look at the cancer genome. The researchers also studied the patterns of genome alterations in cancer, and how DNA repair mechanisms responded.
"This study is yet another demonstration of the power of large-scale whole-genome sequencing," said the senior study author Peter Park, Professor of Biomedical Informatics at Harvard Medical School. "It allowed us to probe the bewildering complexity of genome-shattering in cancer genomes and to characterize common features across hundreds of cases."
"The discoveries made in this project allow us to better understand how cancer arises and evolves, as well as the patterns of alterations in the DNA of human tumors," added Cortés-Ciriano. "Some of these alterations have strong clinical implications and could open new avenues for therapeutic development over the coming years."
This study has suggested that the genome of tumor cells is shaped by chromothripsis; cells jettison suppressor genes and raise the number of cancer-promoting genes to encourage cancer growth. This study can help us learn more about chromothripsis and other cancer genome alterations. These features may also be useful for diagnosing cancer.
Sources: AAAS/Eurekalert! via European Molecular Biology Laboratory, Nature Genetics