A Tiny But Efficient Cas Protein is Discovered in a Bacteriophage
The microbes of the world are locked in a struggle for survival and a battle for resources. They can compete directly in different ways; there are viruses that can infect bacteria, called bacteriophages, and bacterial defense mechanisms that can cut up viral DNA. The gene-editing tool CRISPR-Cas9 is derived from a kind of bacterial immune system. As research on CRISPR has expanded, scientists have looked to other bacteria and found different kinds of Cas proteins that can cut DNA as well as RNA. Now scientists have identified a new kind of Cas protein in bacteriophages.
These new Cas enzymes, called CasΦ (or Cas-phi, because Φ is used to denote bacteriophages), are smaller than the ones that are currently in use, making them easier to get into the nucleus of cells where they edit the genome. One delivery method that's used takes advantage of a harmless virus called adeno-associated virus (AAV). These tiny Cas proteins are now small enough to fit into these delivery systems along with other materials that are needed to perform genome edits. The findings have been reported in Science.
"Adenoviruses are the perfect Trojan horse for delivering gene editors: You can easily program the viruses to reach almost any part in the body," said co-first study author Patrick Pausch, a postdoctoral fellow at the University of California, Berkeley, and in UC Berkeley's Innovative Genomics Institute (IGI).
"But you can only pack a really small Cas9 into such a virus to deliver it. If you would have other CRISPR-Cas systems that are really compact, compared to Cas9, that gives you enough space for additional elements: different proteins fused to the Cas protein, DNA repair templates, or other factors that regulate the Cas protein and control the gene-editing outcome."
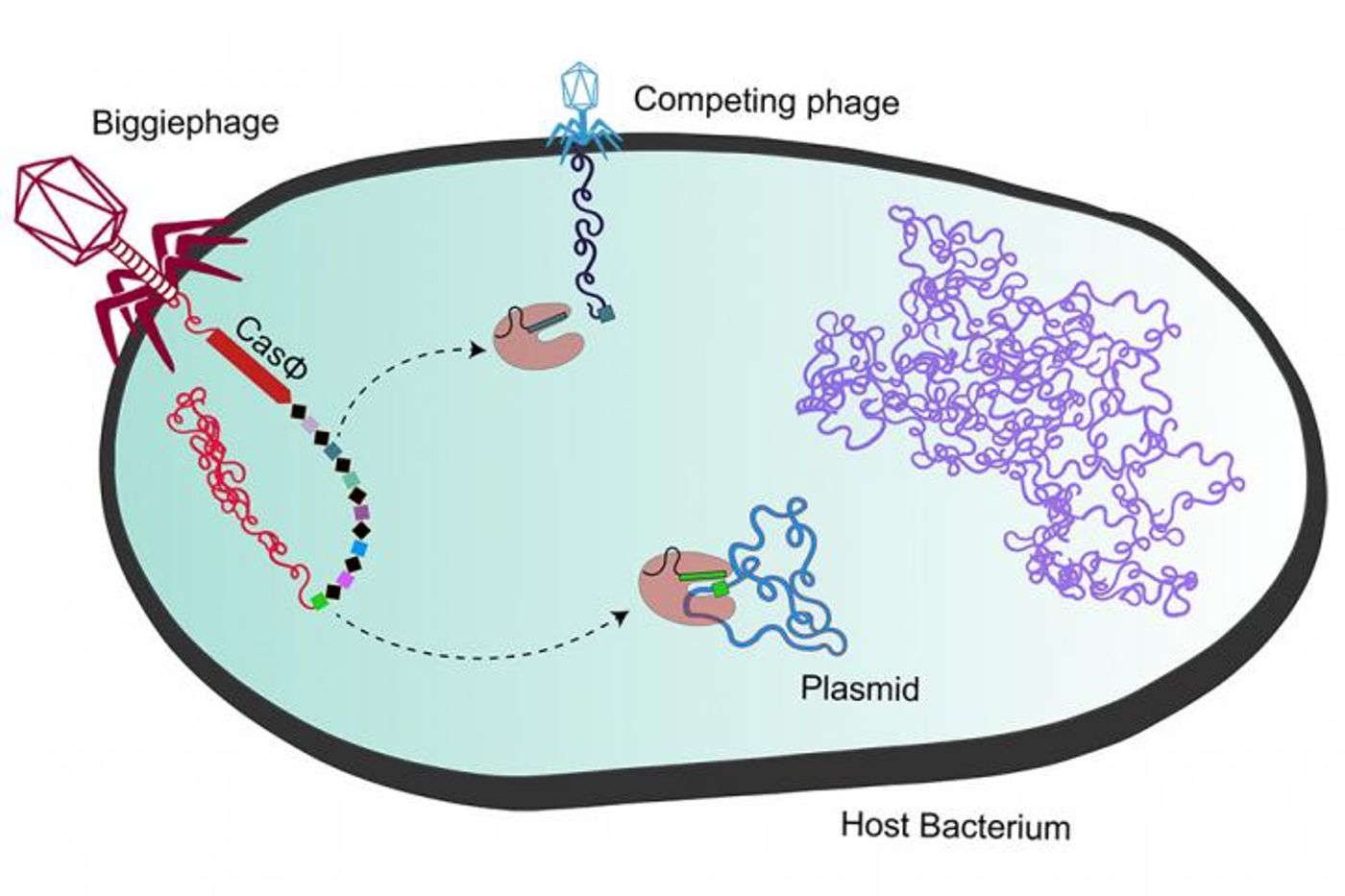
The large bacteriophages or megaphages use CasΦ to manipulate bacteria into battling other viruses.
"The thing that actually made me interested in studying this protein specifically is that all the known CRISPR-Cas systems were originally discovered in bacteria and archaea to fend off viruses, but this was the only time where a completely new type of CRISPR-Cas system was first found, and so far only found, in viral genomes," said co-first study author Basem Al-Shayeb, a doctoral student in the IGI. "That made us think about what could be different about this protein, and with that came a lot of interesting properties that we then found in the lab."
CasΦ is more efficient than Cas9; it has combined functions so it has dispensed with half the enzyme. It's also just as specific and efficient in how it targets DNA as Cas9, and it can work in animal, bacterial, and plant cells.
"This study shows that this virus-encoded CRISPR-Cas protein is actually very good at what it does, but it is a lot smaller, about half the size of Cas9," said IGI executive director Jennifer Doudna, a UC Berkeley professor of molecular and cell biology and of chemistry and a Howard Hughes Medical Institute investigator. "That matters, because it might make it a lot easier to deliver it into cells than what we are finding with Cas9. When we think about how CRISPR will be applied in the future, that is really one of the most important bottlenecks to the field right now: delivery. We think this very tiny virus-encoded CRISPR-Cas system may be one way to break through that barrier."
CasΦ is also able to cut double-stranded DNA, unlike many small Cas proteins that only cut single-stranded DNA. Just as when Cas9 technology emerged, the optimal conditions for the guide RNAs that target DNA sequences are still yet to be revealed, noted Pausch.
Sources: AAAS/Eurekalert! via University of California - Berkeley, Science