Rare Quadruple Helix DNA Found in Live Human Cells
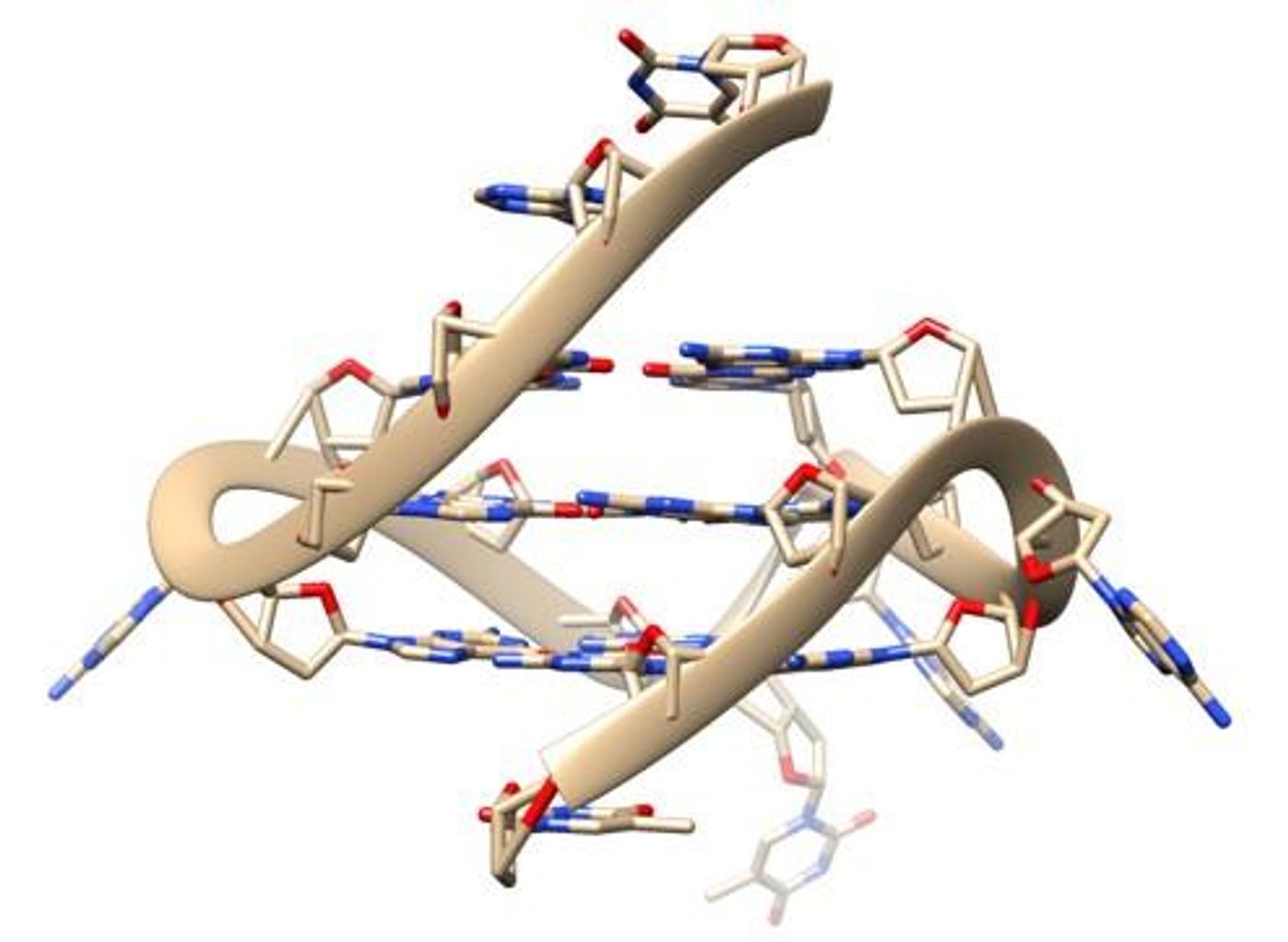
Many people picture the classic double-stranded helix when picturing a molecule of DNA, but DNA is also capable of forming far more complex structures. Usually though, those structures are not observed in live cells. New research has revealed G-quadruplex or four-stranded DNA naturally forming in human cells. The findings, which utilized special probes that reveal how DNA interacts with other molecules, have been reported in Nature Communications.
"A different DNA shape will have an enormous impact on all processes involving it such as reading, copying, or expressing genetic information. Evidence has been mounting that G-quadruplexes play an important role in a wide variety of processes vital for life, and in a range of diseases, but the missing link has been imaging this structure directly in living cells,” explained Ben Lewis from the Department of Chemistry at Imperial College London.
Cancer cells seem to have more G-quadruplexes than normal cells, so they may be playing a role in cancer development. This research revealed proteins that unwind these four-stranded structures, and could potentially be targets for anti-cancer drugs.
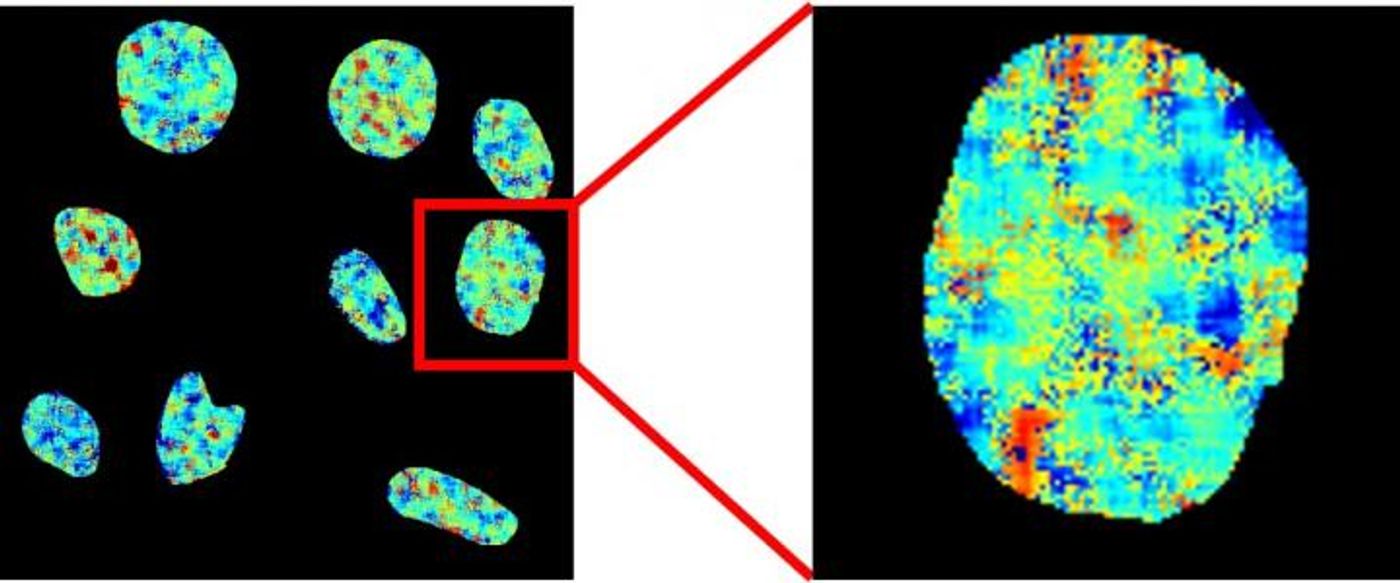
Since G-quadruplexes are rarely observed in living cells, the researchers created a special probe called DAOTA-M2 that lights up when it detects the quadruplexes. In this study, however, the researchers decided to focus on how long the light lasted instead of how bright it was since the probe’s signal does not depend on its concentration or the level of quadruplexes.
"By applying this more sophisticated approach we can remove the difficulties which have prevented the development of reliable probes for this DNA structure," said Dr. Marina Kuimova, of Imperial’s Department of Chemistry.
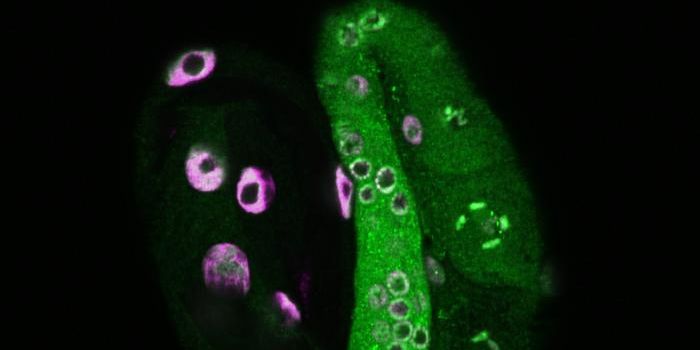
The researchers analyzed how G-quadruplexes interacted with two molecules that unwind the quadruplexes, called helicase proteins. They determined that if the helicases are absent, there were more G-quadruplexes. This suggests that the helicases are involved in breaking the G-quadruplexes down.
"In the past, we have had to rely on looking at indirect signs of the effect of these helicases, but now we take a look at them directly inside live cells,” noted Dr. Jean-Baptiste Vannier of the MRC London Institute of Medical Sciences and the Institute of Clinical Sciences at Imperial.
This study also showed that when a G-quadruplex is exposed to a DNA-binding protein, it will displace the DAOTA-M2 probe and limit how long the probe emits a signal.
"Many researchers have been interested in the potential of G-quadruplex binding molecules as potential drugs for diseases such as cancers. Our method will help to progress our understanding of these potential new drugs," explained Professor Ramon Vilar of Imperial’s Department of Chemistry.
"This project has been a fantastic opportunity to work at the intersection of chemistry, biology, and physics. It would not have been possible without the expertise and close working relationship of all three research groups,” said study co-author Peter Summers of Imperial’s Department of Chemistry.
Sources: AAAS/Eurekalert! via Imperial College London, Nature Communications