Scientists from the University of North Carolina at Charlotte, North Carolina Central University’s
Nutrition Research Program at the NC Research Campus in Kannapolis, NC, and the Carolinas Medical Center in Charlotte, NC have teamed up to identify molecular targets in the signaling pathway for pancreatic ductal adenocarcinoma tumors. Their study was published in the journal
Oncogene last month.
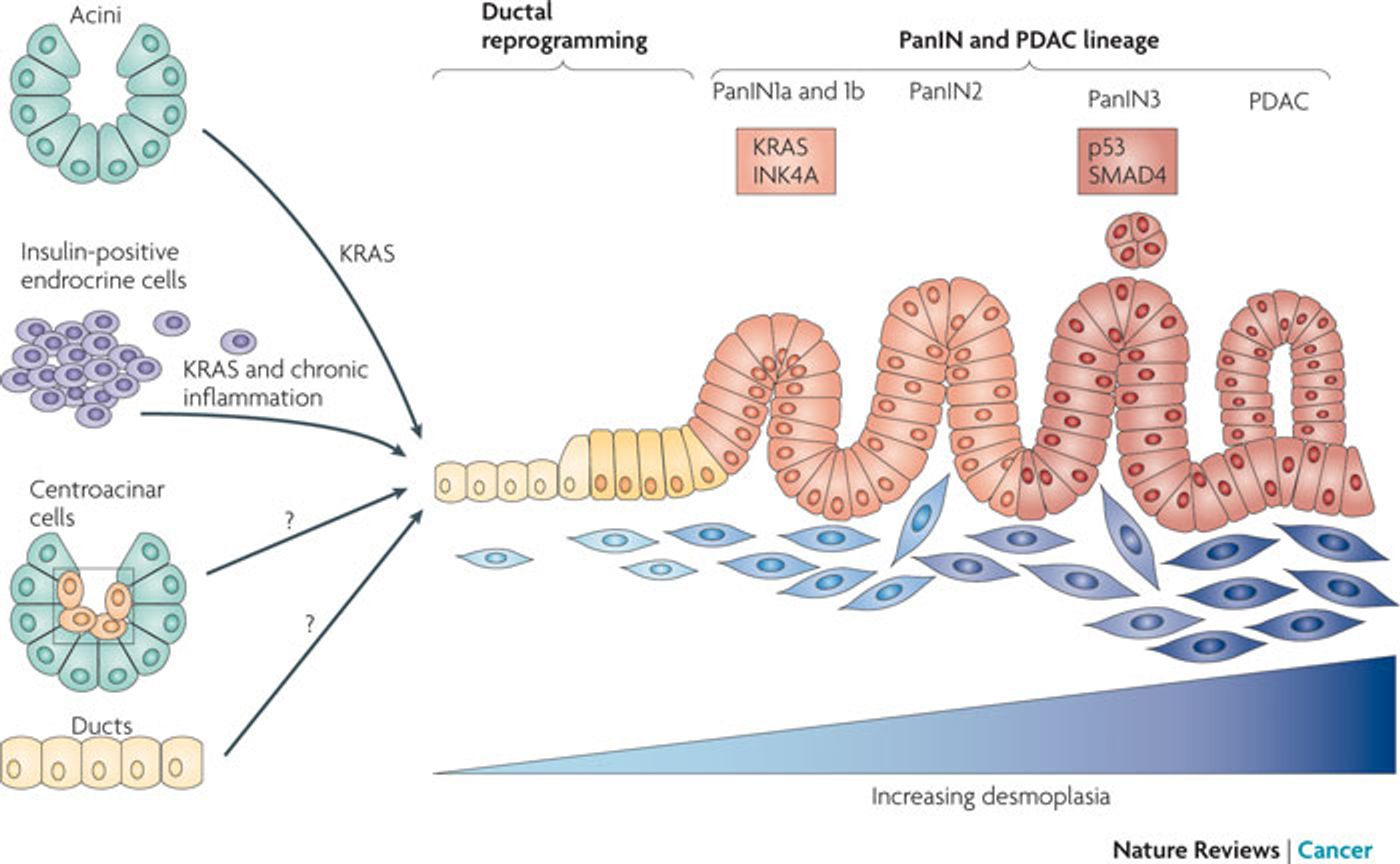
Pancreatic ductal adenocarcinoma (PDA) is a highly lethal type of cancer, with only three to five percent of people surviving five years after diagnosis (
Genes & Development). PDA constitutes about 90 percent of all pancreatic cancers, which was responsible for over 40,000 deaths in the United States in 2015 (
National Cancer Institute). By investigating the key molecular players in the signaling pathway that leads to tumor growth, these researchers may have found a way to block this communication to reduce cancer growth.
Mucin1 (MUC1) is a transmembrane glycoprotein that, in addition to intracellular signaling, is involved in developing protective mucous barriers on epithelial surfaces (
Gene ID). MUC1 is overexpressed in more than 80 percent of PDA cases, which lead the team of researchers from North Carolina to look further at the role of MUC1 in tumor growth.
MUC1 creates a pro-angiogenic tumor microenvironment, forming a network of blood vessels for the abnormally growing tissues to access. MUC1 does so by activating neuropilin-1 (NRP1), also a transmembrane protein and a co-receptor of vascular endothelial growth factor (VEGF) and its ligand.
PDA cells with high levels of tumor-associated MUC1 (tMUC1) and subsequently high levels of NRP1 induce cancer-friendly conditions through signaling with VEGF with a few different actions observed in this study.
- They induce endothelial cell tube formation.
- They generate abnormally positioned blood vessels.
- They enhance distant metastasis (as observed in a zebrafish xenograft model).
PDA cells with tMUC1 also induce N-cadherin and Vimentin, two proteins needed for the epithelial to mesenchymal transition.
The relationship between MUC1, NRP1, and VEGF produces an “attractive therapeutic target” for suppressing PDA tumors, and the researchers from this study uniquely demonstrated that tMUC1 does indeed prompt high levels of NRP1 that lead to a pro-angiogenic tumor microenvironment that does not exist in PDA tumors with low to zero levels of tMUC1. Inhibiting signaling between NRP1 and VEGF suppresses tumor growth, as the team saw in mice models of disease. They did so with an NRP1 antagonist that inhibits the communication between VEGF and NRP1, to cut off all pro-tumor reactions that lead to more lethal cancer growth.
Next, the scientists plan to confirm that inhibiting NRP1 and VEGF signaling in humans produces the same effect of tumor suppression. From there, new therapeutics for PDA can be developed.
Source:
Oncogene