Transcription and translation are two essential life processes for creating proteins, the cornerstone of any Genetics 101 class. Geneticists have been studying how the body makes copies of RNA and translates them into amino acid sequences for protein production for decades, but a new collaborative study published this week in
Nature has produced incredible new images of the transcription process.
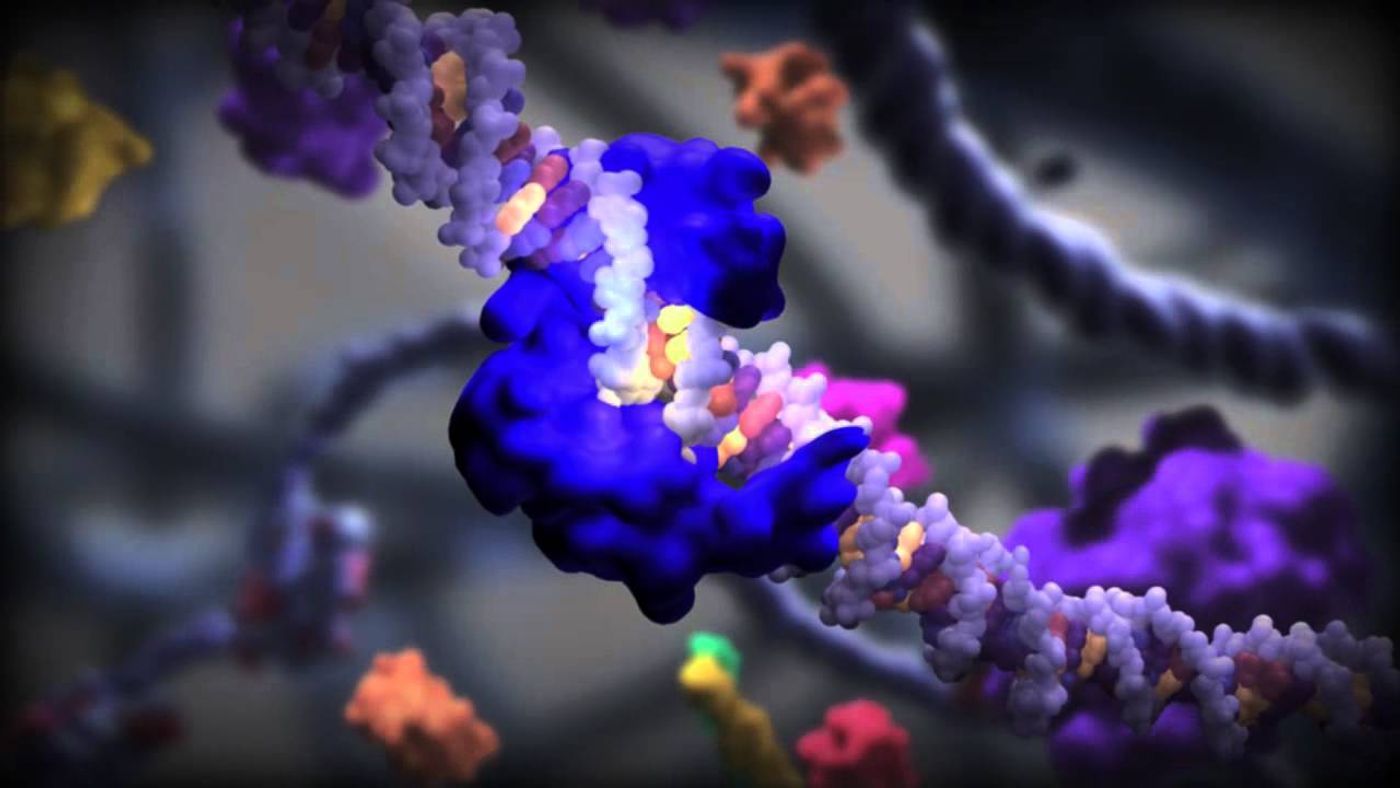
Scientists from Georgia State University, the University of California at Berkeley, and Northwestern University teamed up to observe sequential conformational changes in the pre-initiation complex (PIC) using high-tech cryo-electron microscopy (cryo-EM). This technology is capable of studying samples at cryogenic temperatures, meaning those below the freezing point of water.
“This is a very important system that wasn't accessible by either crystallography or any other structural method before,” said Ivaylo Ivanov, PhD, from Georgia State. “This is the very first near-atomic cryo-EM reconstruction of the human PIC assembly."
PIC is a vital gathering of proteins that facilitate the proper placement of RNA polymerase on the double-stranded DNA helix, prompting the beginning of transcription. The cryo-EM images produced from this study included a never-before-seen complete structure of the PIC, as well as the subunit organization of transcription factor TFIIH, which plays a vital role opening the promoter region in double-stranded DNA for RNA polymerase to begin transcription.
How did they build atomic models of the PIC complex after obtaining the detailed images with cryo-EM? The researchers used “integrative molecular modeling techniques” and “supercomputing technology” to capture and model human PIC starting before transcription began and after messenger RNA was synthesized.
Both observing and imaging critical early events in gene expression for future study is a monumental step forward for the field of genetics, and understanding the “transcription bubble,” as Ivanov calls it, is invaluable for researchers who study genetic diseases and mutations.
Sources:
Georgia State University,
Dr. Ray Radebaugh of NIST’s Cryogenic Technologies Group,
Genome.gov