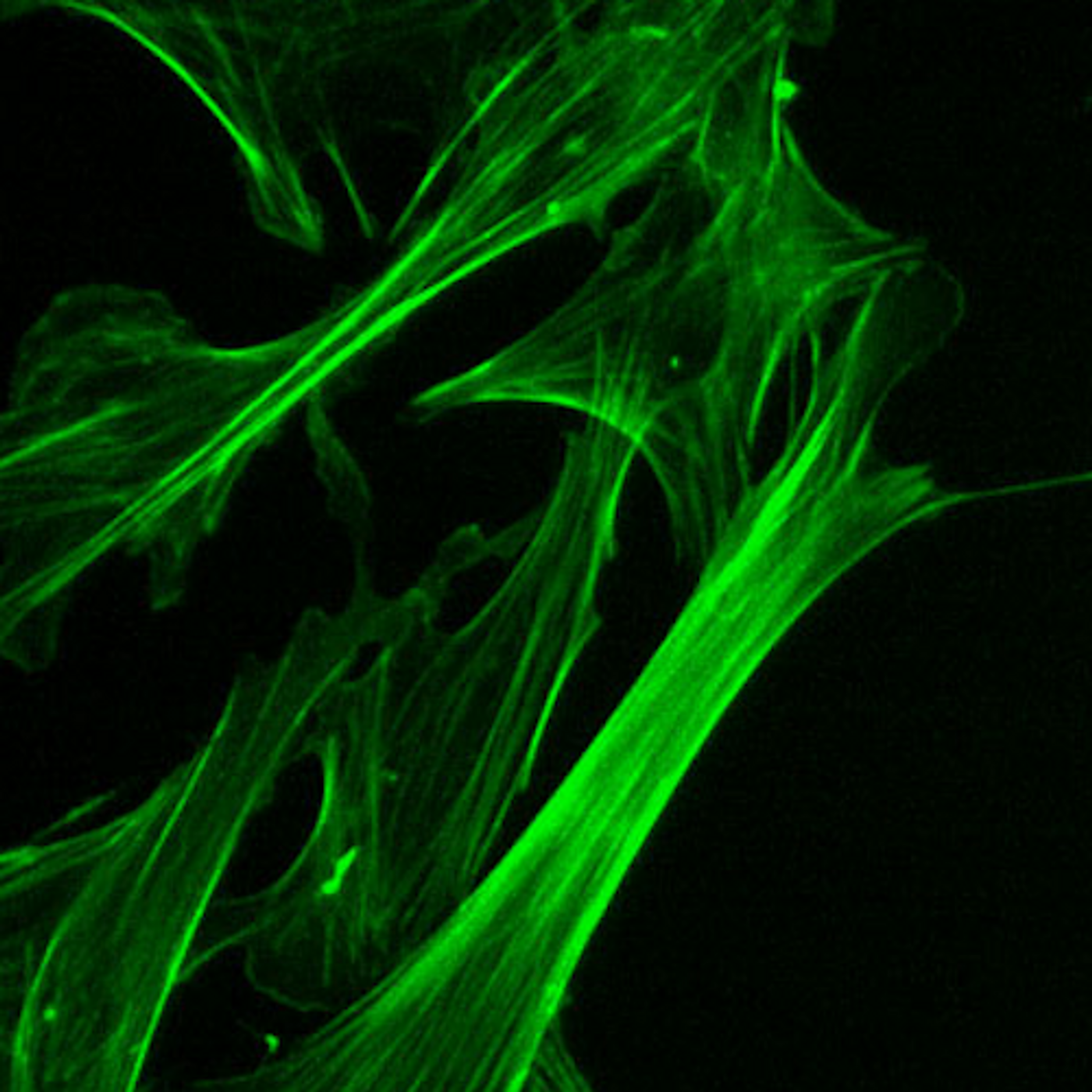
Role of Actin Isoform Determined by Nucleotide, not Amino Acid
There are six different forms of the actin protein in people, and they carry out vital functions. Two are almost the same; they only differ by four amino acids: β-actin and γ-actin. These two proteins don’t perform the same function though, and a long-standing question is why. New data reported in eLife may have found an answer.
"It's a mystery that's been debated in the field for the past 40 years," said Anna Kashina, a professor of biochemistry in the University of Pennsylvania School of Veterinary Medicine. Kashina and colleagues have made an amazing finding. The function of these proteins is determined not by the amino acids they carry, but by the order of nucleotides in their genetic code.
"We like to call it the 'silent code,'" Kashina explained. "Our findings show that the parts of genes that we think of as being silent actually encode very key functional information."
These differences apparently affect the density of organelles called ribosomes, which translate proteins from RNA. Each actin form takes on its own role that is potentially based on that.
Actin is found everywhere, and is known as a ‘housekeeping gene.’ It’s the cell’s most abundant protein. The different forms were once thought of as backups, then research showed that they have their own characteristics and behaviors.
The researchers have built on previous work from their lab which found that a certain modification is exclusive to β-actin because of a change in the coding sequence compared to γ-actin. "We wanted to build on this," Kashina said, "and decided to test the hypothesis, 'What if their functional differences had nothing to do with their amino acid sequence; what if it's all in the genes?'"
Using a the gene-editing technique CRISPR/Cas9, the investigators made changes to only five nucleotides to transform β-actin to γ-actin, even though the variants differ by thirteen percent as ‘silent’ changes that don’t change amino acids. The gene edits worked, and in mice, there was no β-actin protein. However, unlike mice in which the β-actin gene has been knocked out, they were healthy and viable. When the reverse was done with the γ-actin gene, so it only encoded the β-actin protein, the same result was seen. These mice survived, unlike their knockout counterparts.
Later experiments confirmed that the γ-actin proteins which come from an edited β-actin gene would function normally, creating a cytoskeleton and enabling cell migration. "If only the nucleotide sequence is important to protein function, then the mice shouldn't care what protein they have," Kashina said. "And the mice didn't care."
The scientists dug deeper and found that the density of ribosomes on β-actin RNA is over one thousand times higher compared to γ-actin RNA. They learned that every one of the six actin genes had different ribosomal densities. "This suggests that β-actin could translate into protein perhaps a thousand times faster than γ-actin," Kashina said.
As to whether this phenomenon is widespread, the investigators checked protein families with members that are very similar, but are encoded by different genes, and showed changes in ribosomal density. There were many groups shared among different species.
"We think this form of functional regulation is a global phenomenon," Kashina noted. She and her team are continuing the investigation.
The video above highlights some of the different roles of actin isoforms.
Sources: AAAS/Eurekalert! Via University of Pennsylvania, eLife