The Challenges and Advantages of Direct PCR Amplification
Testing for COVID-19 continues to be one of the most important tools in the global fight to slow and reduce the impact of the pandemic, particularly until vaccines are more widely available. Regular screening is likely to become the norm for many people around the world and demand for tests is expected to continue.
Most of the COVID-19 tests are molecular polymerase chain reaction (PCR) assays designed to detect viral ribonucleic acid (RNA) in patient samples. The general workflow for these COVID-19 assays can be broken down into a few steps:
- Collect and store the patient samples
- Ship samples to the testing lab
- Extract genetic material from samples (RNA)
- Amplify target
- Analyze and report the results
In order to expand production capacity and address gaps between global supply and demand, a sample preparation method for direct PCR amplification that simplifies and accelerates the time to results was needed. However, direct PCR amplification comes with both challenges and advantages.
Direct PCR Amplification Challenges
The first challenge is PCR inhibition. Detecting an RNA target requires efficient reverse transcription of the RNA and reliable amplification of the resulting DNA by PCR. Primer and probe annealing should be specific to the intended target and the detection of the amplification signal should be above any background noise.
Sources of PCR inhibition include, but are not limited to:
- The sample itself: e.g., saliva samples contain a high amount of protein which can be inhibitory, blood samples contain hemoglobin, and fecal samples are known to contain a large variety of inhibitory substances
- The sample collection device: e.g., the swab or plastic used to collect the sample
- The sample storage solution: these commonly contain high concentrations of guanidine to inactivate pathogens. However, guanidine is also highly inhibitory to amplification reactions.
In a typical PCR workflow, the solution to minimizing PCR inhibition is to purify the nucleic acid from the sample. If the level of nucleic acid in the sample is high enough, sample dilution can be used to minimize the number of inhibitors in a reaction. But this can impact the sensitivity of low copy targets. Master mix additives like bovine serum albumin (BSA), betane, and trehalose are also commonly used as part of the strategy to overcome PCR inhibitors.
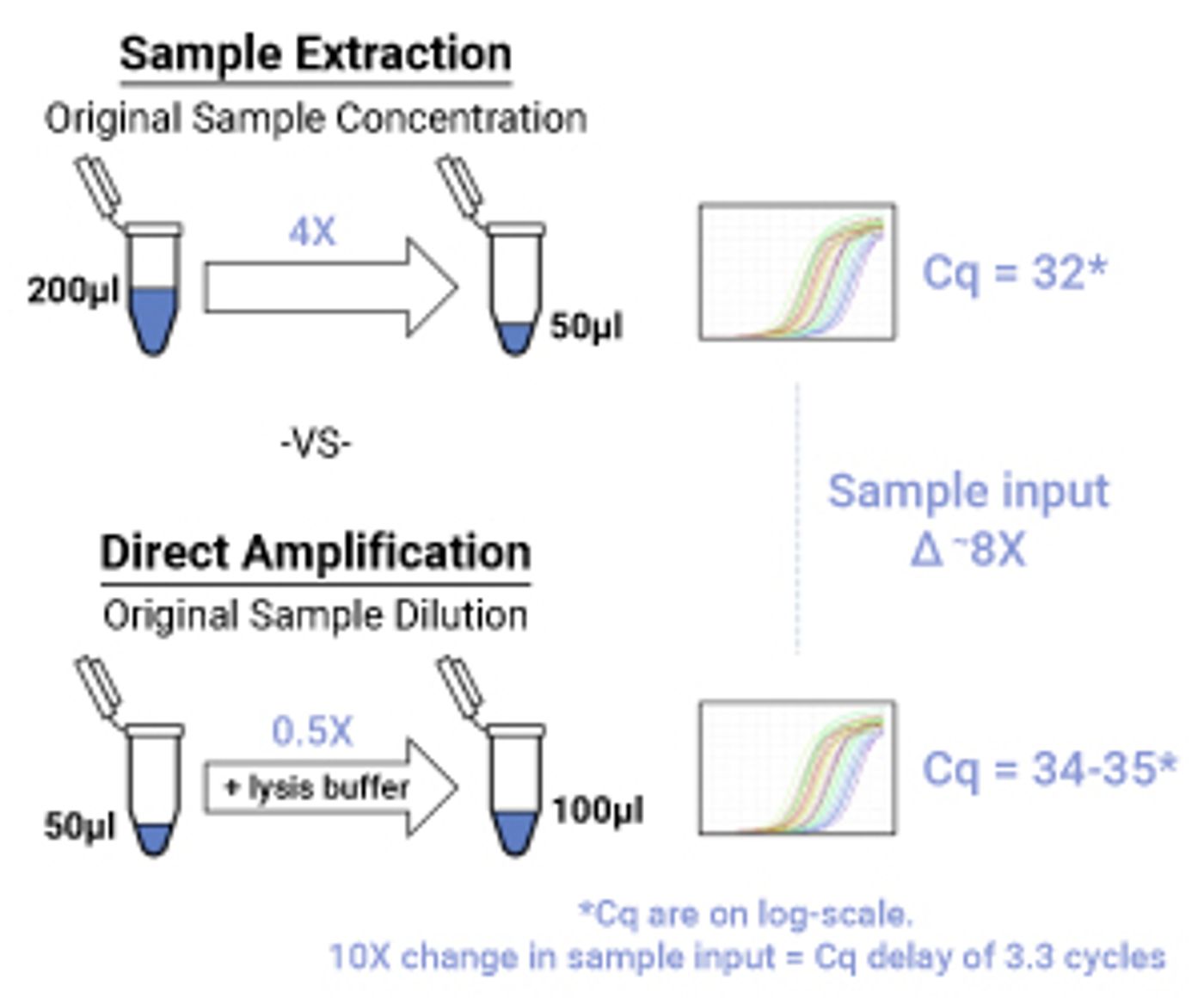
The second challenge to overcome in direct PCR amplification is assay sensitivity. Prior to the sample amplification and analysis by qPCR or RT-qPCR, sample extraction can serve as a concentrating step. In the example in Figure 1, a 200µl sample is concentrated to a 50µl eluate during the purification step. This results in a fourfold concentration increase of the sample before going into the amplification reaction. In direct amplification, there is no extraction step, so there is no concentration. Using the direct amplification protocol for Promega’s XpressAmp™ reagents, the sample is diluted 1:1 with lysis buffer, which results in a twofold dilution, or a 0.5X concentration of the original sample.
Figure 1. Comparison of amplification results of the same sample with a sample extraction workflow and a direct amplification workflow.
When comparing amplification results of the same sample, with the two example workflows, we would expect an eightfold difference in the amplification result. It is important to note that in qPCR, Cqs are on a log-scale, so a tenfold difference between samples would result in a 3.3 Cq difference, assuming 100% PCR efficiency.
Different sample extraction protocols vary in both sample input and elution volume, so it is important to set expectations when comparing a sample extraction workflow with a direct PCR workflow. Typically, most direct amplification workflows would be less sensitive compared to a sample extraction workflow, so additional optimization may be needed when converting an assay to a direct PCR workflow.
Direct PCR Amplification Benefits
Despite these challenges, there are key advantages to direct amplification. In a typical PCR workflow, the purification wash steps utilize many reagents and buffers, often supplied in configurations that are dependent upon plastic consumables and specific instrumentation. In a direct amplification workflow, you circumvent all those steps, reagents, and plastics, allowing you to avoid potential throughput limitations from different supply constraints.
Utilizing a direct PCR workflow can also help you save time in the lab, though the amount of time saving will vary depending on your previous extraction method, and whether it was automated or manual. Direct amplification workflows are also high throughput automation friendly across multiple platforms.
Learn more about direct PCR amplification solutions and how they can be customized to fit your needs.
About Promega Corporation
Promega Corporation is a leader in providing innovative solutions and technical support to the life sciences industry. Our product portfolio of over 4,000 products support a range of life science work across areas such as cell biology; DNA, RNA and protein analysis; drug development; human identification and molecular diagnostics. For over 40 years these tools and technologies have grown in their application and are used today by scientists and technicians in labs for academic and government research, forensics, pharmaceuticals, clinical diagnostics and agricultural and environmental testing. Promega is headquartered in Madison, WI, USA with branches in 16 countries and over 50 global distributors. For more information, visit www.promega.com.