Auto-antibody Detection for Rheumatoid Arthritis Patients
No case of rheumatoid arthritis (RA), an autoimmune disease, is the same. Now, researchers want RA diagnostic approaches to match its pathological diversity. From Uppsala University, scientists demonstrate the importance of a broad diagnostic approach to identifying cases of RA.
“RA patients with and without ACPA probably have different diseases with separate causes, but start with a similar clinical picture," explained research team leader Johan Rönnelid.
ACPAs, or anti-citrullinated protein antibodies, are produced by the immune system as part of the autoimmune response present in RA. Autoimmune diseases affecting different body tissues and systems are all caused by the body’s accidental attack on its own tissues, responding as if the tissues were pathogenic. A key part of this autoimmune response is the production of “auto-antibodies,” like ACPAs, that target the body’s own proteins, like the amino acid citrulline, as opposed to targeting bacterial or viral proteins.
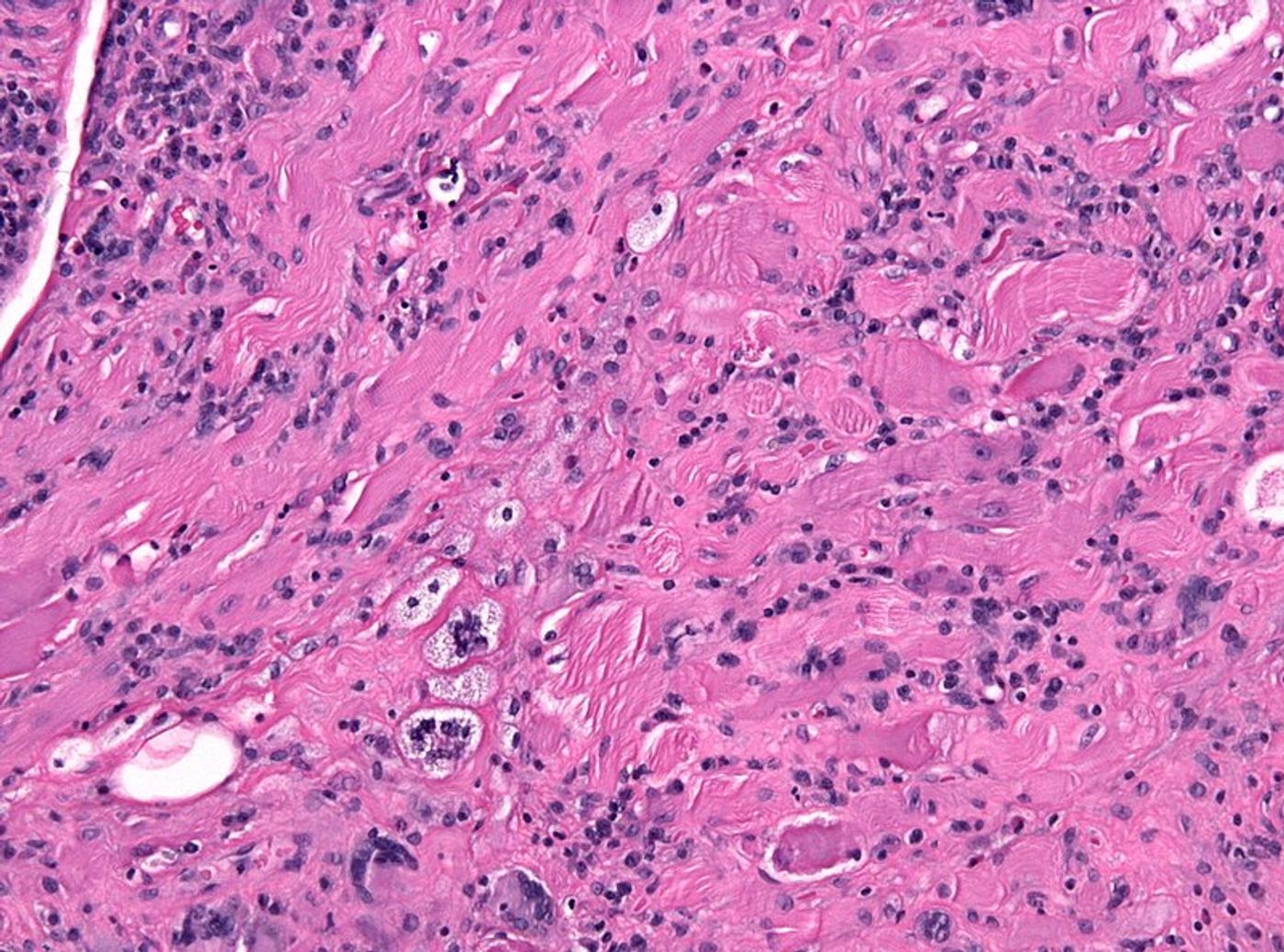
RA is an autoimmune disease characterized by chronic inflammation in the joints and other parts of the body. Inflammation in the joints causes tissue to thicken, resulting in swelling and pain experienced by RA patients. The disease often affects the hands, feet, wrists, elbows, knees, and ankles. Approximately 1.5 million people are diagnosed with RA in the United States, disproportionately women.
Researchers from the present study are dedicated to improving RA diagnosis with a new technique that measures a variety of citrulline antibodies. Looking for a variety of citrulline antibodies as opposed to one type or just a few types will help scientists detect severe inflammation and joint damage.
A broader test is necessary to detect the various proteins and protein fragments that are targeted in each case of RA. No case is the same, so the test has to account for all of the possibilities. The test looks for various auto-antibodies, which bind to host proteins and form immune complexes that then trigger inflammation that leads to the physical symptoms experienced by RA patients. The current study is the first to observe ACPA immune complexes in body fluids from RA patients.
ACPAs are produced as a result of RA-mediated inflammation, and they are observed in more than half of all RA patients, but not all. In the past, researchers have identified genetic differences between patients with and without these antibodies. Additionally, ACPA-positive RA cases are linked to patients who smoke, and ACPA-positive patients have more joint inflammation than ACPA-negative RA patients.
Researchers developed the new test by first extracting immune complexes from different types of body fluid (serum, synovial fluid, immune complexes) from 77 RA patients, then measuring 19 different ACPAs. They found that no single ACPA stood out from the rest. In fact, a broader range of ACPAs in immune complexes are responsible for driving inflammation and joint damage characteristic of RA.
The present study was published in the journal Annals of the Rheumatic Diseases.
Sources: Arthritis Foundation, Uppsala University