The Immune Systems Molecular Alphabet
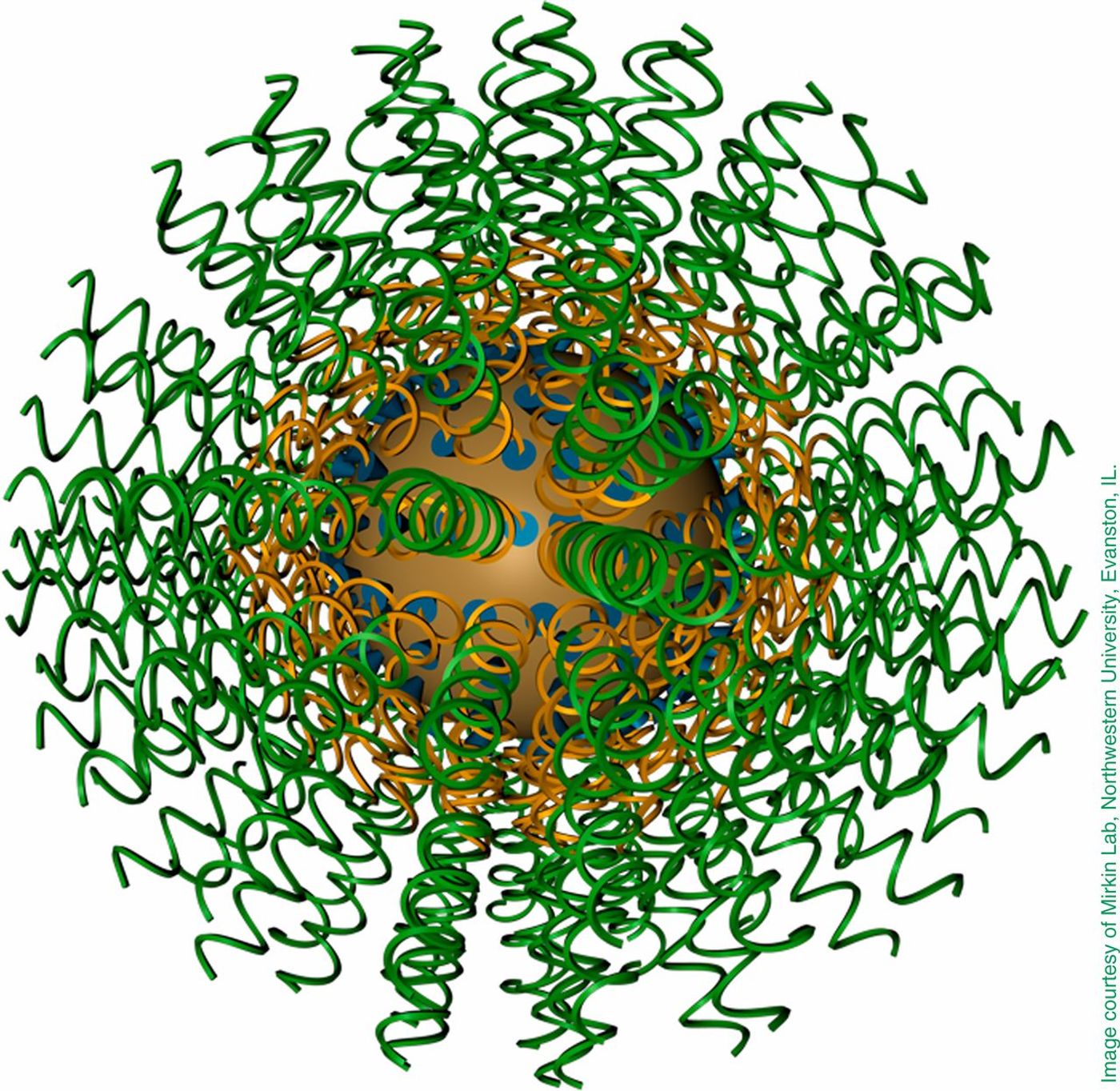
Therapeutic nucleic acids (TNAs) include a wide range of materials made of RNA, DNA, and their analogs that have the potential for disease detection and treatment. These TNAs hold promise for drug therapy to treat diseases such as cancer, diabetes, infectious disease, and cardiovascular disease among many others. A subset of TNAs, the Nano-TNAs, includes programmable nucleic acid nanoparticles (NANPs) that include self-assembling nucleic acids serving as building blocks for novel entities. The technology of NANPs is relatively new but shows promise for biological materials and medical applications. NANPs are programmable and self-assembling and open to chemical modification, control over functionalization, predictable and regulatable, and consistent batch-to-batch. Despite advancements in basic research, clinical trials have not been reached with NANPs yet.
A recent study from the Nanotechnology Characterization Lab at the Frederick National Laboratory for Cancer Research tested the immune effects of NANPs and measured responses from various immune cells. The immune system responds to stimuli such as nucleic acids, antibodies, and small molecules leading to adverse effects or desirable activation of the immune response that is beneficial to the host. The ability to control the response of the immune system to these stimuli can avoid harm or promote the beneficial immune response depending on the given situation. Previous studies on the use of TNAs to affect gene or cell function resulted in extreme side effects, provoking a harmful and potentially fatal reaction from the immune cells. Use of designer self-assembling nanoparticles that combine the effects of several sequences into a targeted drug, creating multiple effects in one particle, was assumed to give the same toxic effect as the natural nucleic acids.
To test the toxicity of the designer particles, the study published in Nano Letters, utilized 25 different DNA or RNA nucleic acid nanoparticles that had been designed by researchers in the nanoscience field. The particles varied in shape, size, molecular weight, and critical chemical attributes. The blood of 60 unique human donors had particles introduced; the samples were then analyzed for production of 28 different cytokines. Results showed that “naked” NANPs, unattached to other biological molecules, elicited no immune response since they are unable to enter any immune cell alone. Paired with carrier molecules, the NANPs entered immune cells and elicited a clear response. The immune response varied based on particle size, shape, structure, DNA vs. RNA, and chemical nature instead of eliciting a uniform response as hypothesized. DNA particles caused less immune response than RNA, while the difference in RNA shape showed that RNA cubes elicited more of an immune response than RNA rings or fibers. The cytokines produced differed based on shape as well as RNA or DNA, with DNA cubes inducing type I interferons alpha and omega and RNA cubes inducting type I interferon-beta or type III interferon-lambda.
The difference in particle design and immune response creates an “alphabet” of sorts that could be used to invoke an immune response overall or trigger a precise immune response. Kirill Afonin, one of the studies corresponding authors, said of the studies future applications "If you need to deliver a drug, you may want the carrier to be non-immunogenic. We can tell you exactly which particle you can use for that, but if you want to stimulate the immune response, for example, to activate the immune system against cancer. Then you can use certain particles which will activate the immune response but avoid inflammation. We can produce interferons, but not inflammatory cytokines, for example. This is like sharpshooting," he explained. "You will be shooting for a particular cytokine, without touching others. This is like a letter or a word, like a text message that we send to the immune system. The immune system will read your message and text back with the interferon."
To explore more about well-known therapeutic nucleic acids, watch the video below on a traditional TNAs, the siRNAs.