Yin & Yang: The Duality of Cancer-Associated Fibroblasts in Pancreatic Cancer
Pancreatic Cancer is a devastating disease. This year alone, fifty-five thousand new patients were diagnosed in the United States (1). Unfortunately, not only is it painful, but it is usually diagnosed too late for effective treatment with currently available therapies. Researchers in the Tuveson Lab at Cold Spring Harbor Laboratory are hoping to change the fate of those diagnosed with pancreatic cancer by targeting the cancer-associated fibroblasts (CAFs) that support tumor growth.
Pancreatic Ductal Adenocarcinoma (PDAC), the most common type of pancreatic cancer, has a 1-year survival rate of 20% and a 5-year survival rate of less than half that (2). Prognosis is so poor because treatment is complicated by the lack of blood vessels in the tumor and the high rate of metastasis. Without a blood supply, chemotherapies can’t reach or target cancerous cells, and PDAC’s location near ducts allows cancerous cells to slough off and establish satellite tumors at other locations.
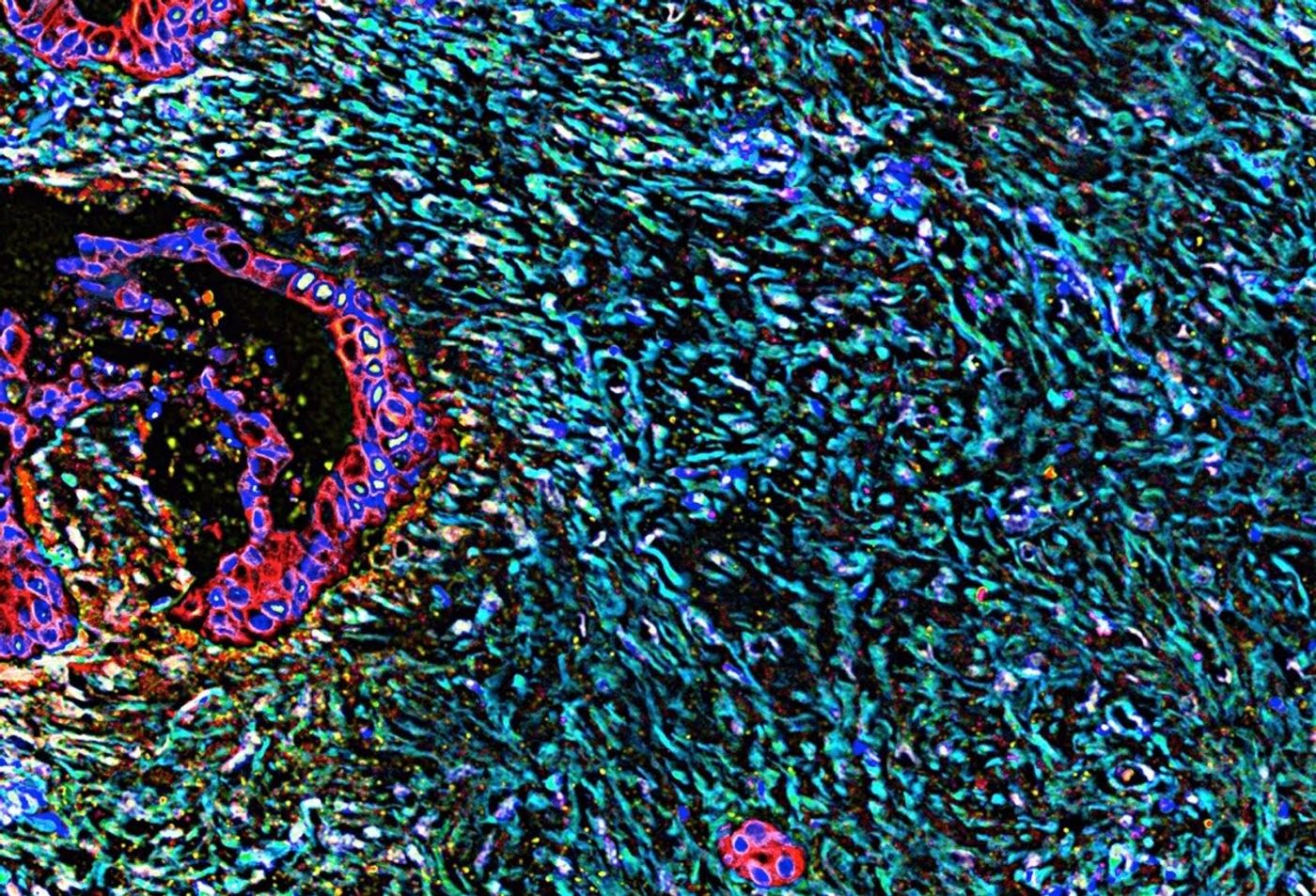
Like all solid tumors, PDAC is made up of both cancerous cells and a support system, the stroma (3). However, PDAC is unique in that close to 90 percent of the tumor mass is stroma, and it is in the stroma that CAFs secrete factors that promote tumor progression and metastasis. (4).
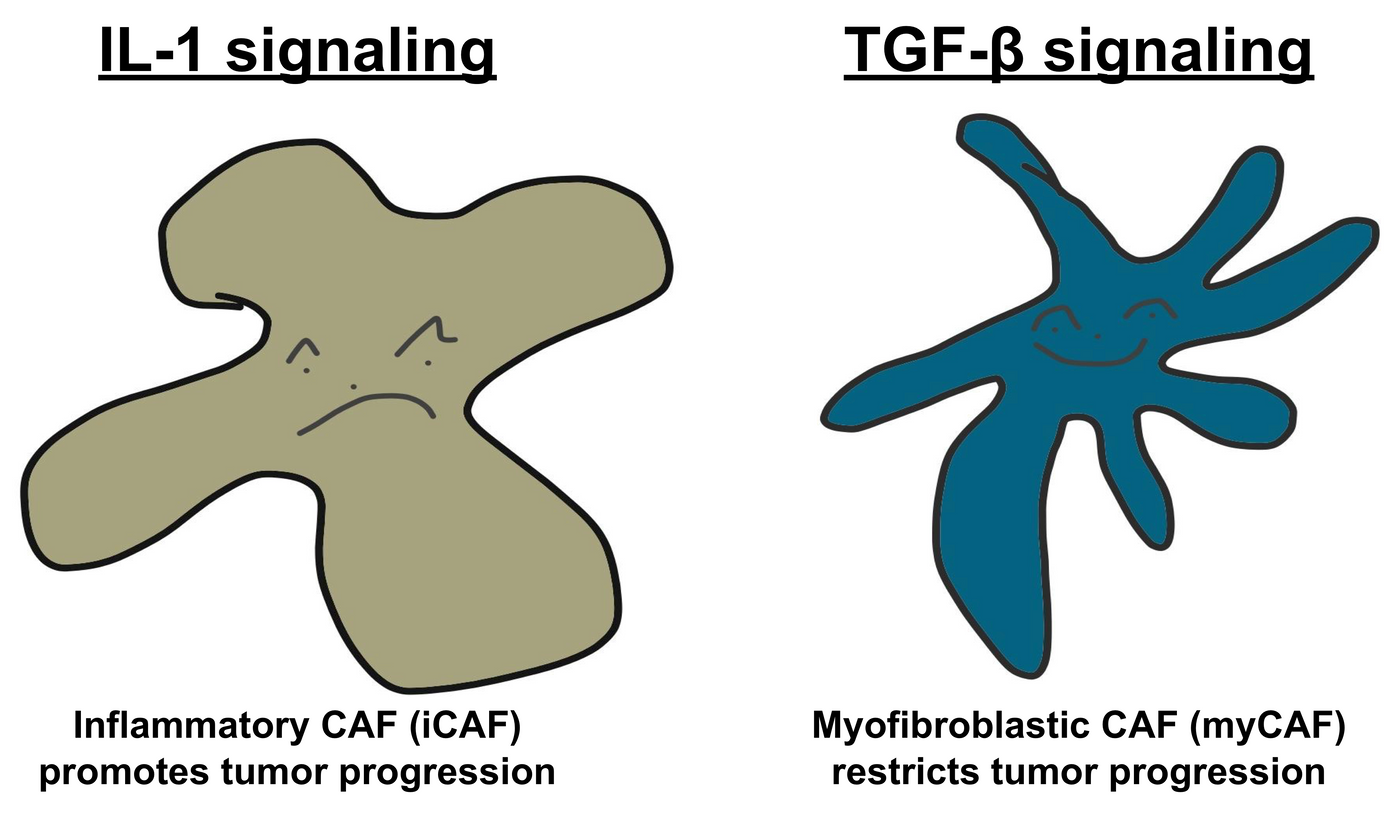
Previously, CAFs were found to be pro-tumorigenic. However, studies and clinical trials designed to target CAFs failed to inhibit the progression of PDAC, and in some cases exacerbated it (Biffi et al). This suggests that there must be heterogeneity in the CAF population which is exactly what Biffi et al saw. They demonstrated that PDAC stroma contains two subsets of CAFs, one inflammatory (iCAF) located close to the cancerous cells, and the other myofibroblastic (myCAF) located more distally.
In this study, they built on that observation and determined the factors which control the differentiation of each type of CAF. Following single-cell RNA sequencing of both iCAFs and myCAFs, they used biochemistry and learned that while IL-1 signaling turns on pathways in CAF precursors that cause differentiation into iCAFs, TGF-β downregulates the IL-1 receptor (IL-1R) resulting in those precursors adopting a myofibroblastic phenotype (myCAFs).
But they didn’t stop there. Using a mouse model that recapitulates PDAC, they inhibited signaling downstream of the IL-1R and not only saw a significant reduction in tumor burden, but the small, remaining tumors contained more tumor suppressive myofibroblastic CAFs.
So, the previous clinical trials that inhibited CAFs may have failed because they inhibited both the inflammatory iCAFs and the tumor-suppressive myCAFs. It is possible that by specifically targeting iCAFs in patients’ pancreatic cancers, a diagnosis of pancreatic cancer will no longer be a death sentence.
Learn more about cancer-associated fibroblasts from the video
Sources: 1) cancer.org; 2) pancreatic.org; 3) Journal of Thoracic Oncology; 4) Genes & Diseases; 5) Journal of Experimental Medicine