A Fatty Diet can be Fuel for Tumor Growth!
Obesity is a significant problem in our societies with increasing the risk for many diseases, including cancer. Cancer is affected by obesity and metabolic changes, but the exact process by which obesity affects cancer was not clear.
A recent study that has been done by Harvard medical school and published in Cell investigated the obesity-cancer relationship. They found that cancer cells rely on a high-fat diet, associated with obesity, for fuel and compete with immune cells for it. CD8+ T cells, which are essential for fighting and killing tumor cells, and are the focus of cancer immunotherapies, get depleted of fats by cancer cells, which decrease their number and activity.
“Putting the same tumor in obese and nonobese settings reveals that cancer cells rewire their metabolism in response to a high-fat diet,” said Marcia Haigis, professor of cell biology in the Blavatnik Institute at HMS and co-senior author of the study. “This finding suggests that a therapy that would potentially work in one setting might not be as effective in another, which needs to be better understood given the obesity epidemic in our society.”
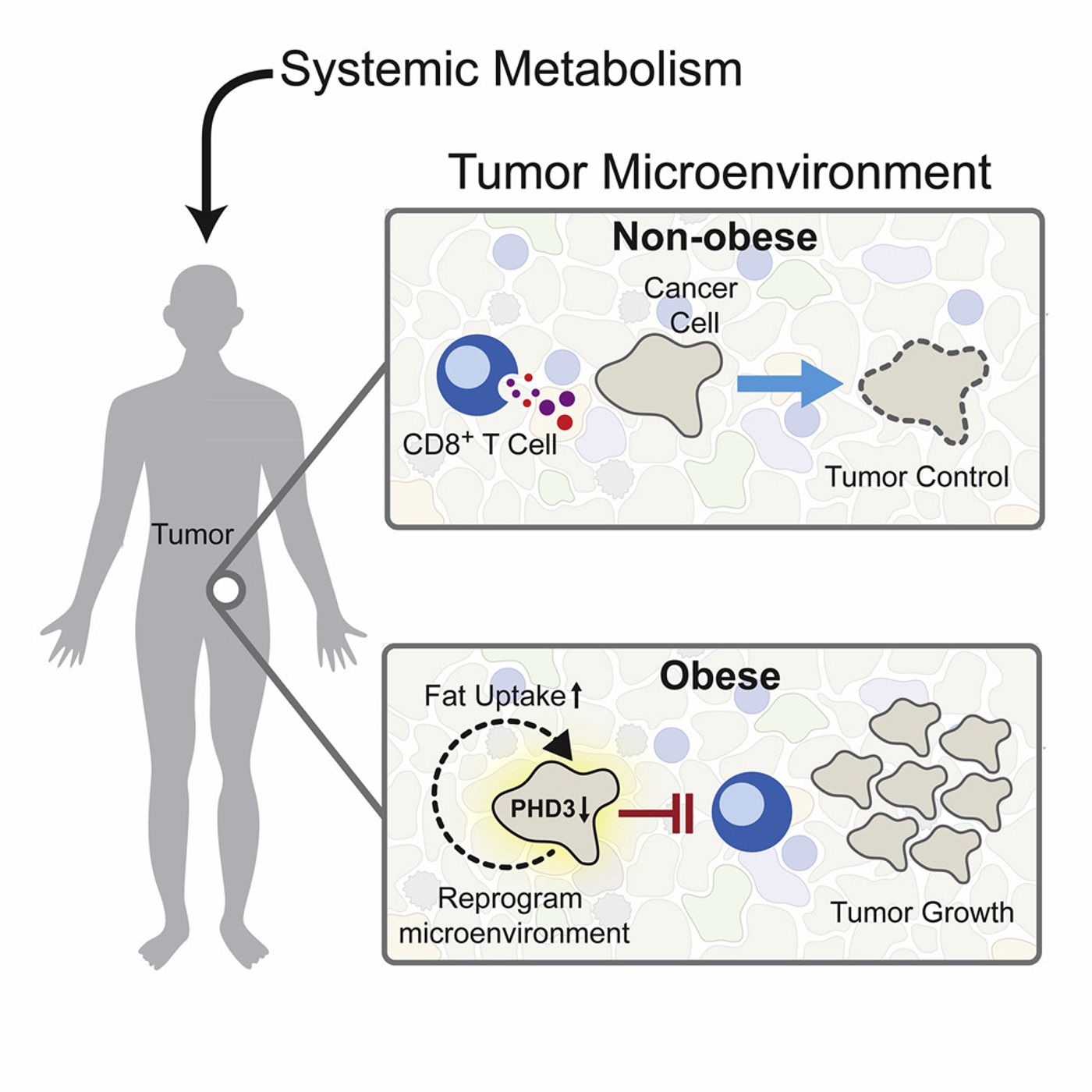
They studied the tumor microenvironment and compared the changes in mice given high-fat and regular diets. They found out that tumors grew rapidly in obese mice compared with the ones having a regular diet. The activity and number of CD8+ T cells decreased but only in the tumor microenvironment and not elsewhere in the body. These diet-related changes didn't affect tumor growth when CD8+ T cells were experimentally removed in mice. Cancer cells underwent metabolic changes in obese mice to increase their uptake of fat for fuel, but CD8+ T cells did not, which made them starve.
Credit: Cell Journal
Those changes were only visible in immunogenic cancers, which can induce an immune response and have a high number of immune cells.
Also, the tumor microenvironment was depleted of essential fatty acids used as fuel by immune cells, although they were present in the rest of the body.
Another observation was that cancer cells in obese settings had lower expression of a protein that is used to stop excessive fat metabolism, "PHD3" compared to in normal settings.
When the researchers increased the expression of PHD3, it reversed the negative effect of obesity on immune cells. The tumors grew slower than in cells with lower PHD3 expression. This result was due to increased CD8+ T cells activity as increased PHD3 expression stopped the cancer cells from taking up fats and increased essential fatty acids necessary for immune cells. Again those changes were not visible in mice lacking CD8+ T cells.
“CD8+ T cells are the central focus of many promising precision cancer therapies, including vaccines and cell therapies such as CAR-T,” Sharpe said. “These approaches need T cells to have sufficient energy to kill cancer cells, but at the same time, we don’t want tumors to have fuel to grow. We now have amazingly comprehensive data for studying these dynamic and determining mechanisms that prevent T cells from functioning as they should.”
These findings could open the door for new therapeutic targets and strategies to fight cancer and increase the efficacy of immunotherapies.
Sources: ScienceDaily via Harvard Medical School