A Potential Treatment Approach for Crohn's Disease
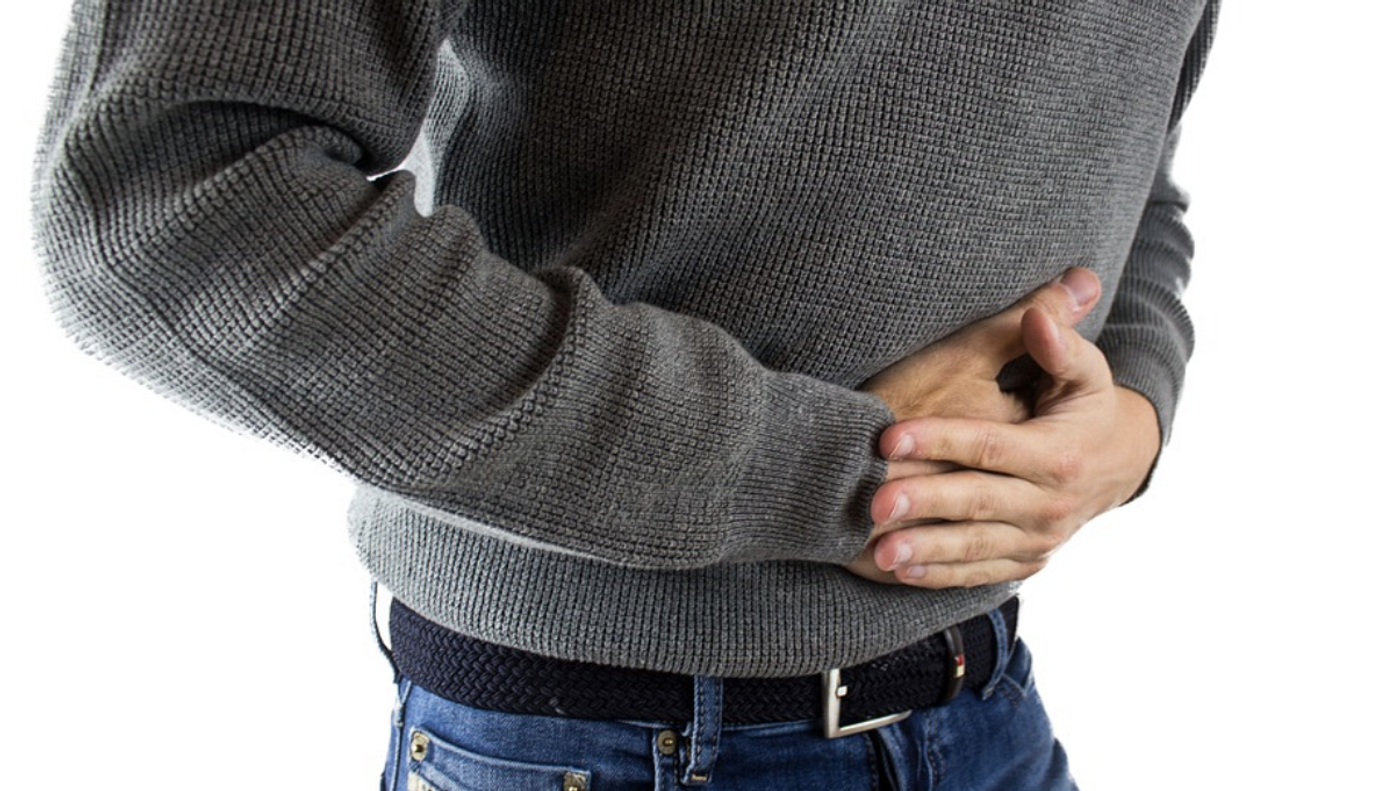
Researchers may have found a way to treat or prevent Crohn's disease, one of the most common forms of inflammatory bowel disease. New work showed that if a particular subset of immune cells were removed and another type was increased, colitis could be prevented in both a mouse model and in patient cells growing in culture. The findings, which could lead to the development of a treatment for Crohn's, have been reported in Science Immunology.
In inflammatory bowel disease, the immune system overreacts to the community of gut microbes that reside naturally in the gastrointestinal tract, called the gut microbiome. One reason for this is a protein called flagellin that composes the flagella found on some bacteria, which helps them move around. Short-lived T effector cells can go into overdrive because of flagellin.
The flagellins found in the Lachnospiraceae family of bacteria, such as one called CBir1, are especially immune-reactive. More than half of Crohn's disease patients have been found to have immune overactivity in response to CBir1 and related flagellins.
Another type of immune cell, T memory cells (Tm), retain a memory of previous encounters with cells or substances they've identified as invaders, and they can recall encountering flagellins. They will remain quiet for long periods unless they meet the flagellin protein again, in which case the Tm cells are reactivated. That reactivation will change the cells' metabolism and trigger their proliferation into huge number of T effector cells. A signaling protein known as mTOR is essential for this transition from inactive to active.
Since mTOR is also involved in the initial encounter between T cells that are naive and flagellin, the researchers were interested in inhibiting mTOR to stop the activation of these immune cells, at the same time that the cells were being stimulated by flagellin antigens. They achieved this by generating a synthetic peptide carrying several repeats of one CBir1 epitope; it activated the memory cells but did not trigger an innate immune response. Already-existing drugs can act to halt mTOR.
Research leader Charles O. Elson, M.D., professor of medicine at the University of Alabama at Birmingham calls this strategy cell activation with concomitant metabolic checkpoint inhibition (CAMCI).
When CAMCI was used in a mouse model, there were reductions in antigen-specific CD4-positive T cells and memory responses. Colitis was prevented in a mouse model and similar effects were seen in cell lines from Crohn's patients by using this strategy.
In a therapy for Crohn's patients, Elson noted that more than one flagellin would likely have to be targeted to have a good effect. "...we anticipate the future use of a synthetic multi-epitope peptide containing multiple CD4-positive T cell flagellin epitopes to target many microbiota-flagellin-reactive CD4-positive Tm cells," Elson said. "Depending on the serologic or CD4-positive T cell response to certain microbiota antigens, this CAMCI approach could be tailored to individuals with different combinations of epitopes as a personalized immunotherapy."
Intermittent application of CAMCI could be useful for helping Crohn's patients stay in remission, said Elson. "And with autoantigen epitopes better studied in the future, this approach could be expanded to treat other inflammatory or autoimmune diseases such as type 1 diabetes or multiple sclerosis."
Sources: AAAS/Eurekalert! via University of Alabama at Birmingham, Science Immunology